Dental Lasers and the Dental Hygienist
Is anyone else out there confused about dental lasers? I know I was when my doctor asked me if I knew of any periodontal lasers or of anyone using them. He wanted to incorporate laser dentistry into our practice for our periodontal patients. I felt really ignorant since I had no idea how to answer his question. I should know these things; I am the hygienist in the office — the periodontal expert!
So I started out on a mission to find all of the information I could about periodontal lasers and their uses. Little did I know what was in store for me and how much information there is to learn about laser dentistry! It was all so confusing at first when I read articles discussing light spectrums, wavelengths, and physics. So I created this article to simplify things and give you a real-life perspective of lasers.
Lasers are revolutionizing the dental and medical world as I write. Laser dentistry is fascinating, but for many of us it is confusing — access to information is limited because the cutting-edge technology is so new. From what I have seen and learned so far, lasers may just revolutionize the way we practice dentistry and change the way we approach periodontal disease as a whole.
Interest in lasers has been gaining in recent years, and it is estimated that 4,500 surgical dental laser systems are currently in circulation in the United States.9 That means of the 140,000 practicing dentists in the United States, 3% to 4% of them are using laser technology. Laser use is on the rise. If we do not learn the applications for lasers, we may find ourselves left out of the loop or, even worse, losing our patients to practices that are keeping up with the times.
If you are just starting to investigate laser technology and its application to your practice, it can be difficult and confusing. For most of us, laser technology was not even around when we were in dental hygiene school. Even new graduates are not being educated on laser use in the dental community. This article will attempt to simplify laser technology for you as well as provide valuable, practical information for you to be able to apply in your own private practice setting.
How Lasers Found Their Way Into Dentistry
What exactly is a laser? The word laser is an acronym coined in 1960 and stands for: (L)aser (A)mplification by (S)timulated (E)mission of (R)adiation.19 A laser is a powerful beam of light that can produce intense heat when focused at close range. It concentrates high energies into an intense, narrow beam of nondivergent, monochromatic electromagnetic radiation of one wavelength.19 (Definitions of various terms in this article are found in Table 1.)
Lasers were first postulated by Albert Einstein in 1916. He suggested that portions of the electromagnetic field could be stimulated, thus producing amplified light. In the 1950s and 1960s, many advances were made in the field of laser technology that led to the types of lasers we now use today. In 1958, two physicians named Charles Townes and Arthur L. Schawlow discovered how to make a device that would emit visible light, not just microwaves, and they called their discovery a "Master Optic."20 The first laser was made by this mechanism in 1960 by physicist Theodor Maiman. Then, in 1961, W.R. Bennet and D.R. Heriott developed the first helium-neon laser. In 1964, C.K.N. Patel made the first CO2 laser, and in 1968 the first Nd:YAG (neodymium-doped yttrium aluminum garnet) laser was completed, which are both used in dentistry today.20
Lasers were first used in biology studies and research, then later in medicine. Laser use in dentistry dates all the way back to 1964 and starts with Stern and Sognnaes.20 They were the first inventors to look at lasers for applications on hard dental tissues. They found ruby lasers could reduce the permeability of exposed enamel to acid demineralization. Since this time laser use has continued to grow in the dental field and the applications have spread to soft tissue use as well.
The first soft tissue laser use was in 1985 by a pioneer in the field of lasers named Pick, an oral surgeon.20 He started using CO2 lasers for gingivectomies. Until recently, soft tissue applications of lasers has been mainly confined to oral surgery; but with the new technology that was developed in the 1980s, lasers on the market today now treat periodontal disease. Many periodontists and even general dentists are utilizing this laser technology and watching their practices thrive from its results.
The Physics of Lasers
The use of lasers in your practice will be discussed in this article but let's give you information about laser physics and mechanisms first so you can better understand how lasers work.
Lasers are a single color of light; they are a man-made single-photon wavelength. They differ from "white" sunlight, which is a continuum of light of many different wavelengths such as visible colors, infrared (heat), and ultraviolet wavelengths.15 Laser light is produced when an excited atom (see Table 1) becomes stimulated, thus emitting a photon (see Table 1). This generates a coherent, focused, concentrated monochromatic (see Table 1)ray of light which will allow us to have a directed effect on our target tissue at much lower energy levels than if we were to use natural light.4,13
The overall effect of laser light on its target is dependent on its wavelength (see Table 1). Different wavelengths are absorbed at varying rates and this corresponds to their depth of penetration; thus enabling some lasers to have greater precision and accuracy than others.11 Later in this article, I will discuss individual lasers and their applications, and you will see how these different wavelengths influence the function of the laser (see wavelength table).
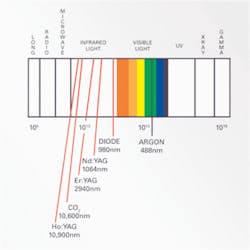
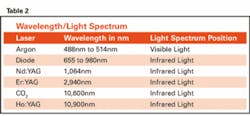
It is also important to understand light when learning about lasers. Where a laser lands on the light spectrum (see Table 1) greatly influences its properties (see light spectrum table).
You will see that most of the dental lasers in use today fall into the infrared zone. In the infrared zone, laser beams have very short wavelengths that are shorter than visible light. This means less energy is needed to create the single color laser light. The only laser in the visible light spectrum is the argon laser, and you will later see why that is. The argon laser is not used for the same dental procedures as the infrared lasers. The general rule of thumb here is the higher the laser goes into the infrared (Ho:YAG and CO2), the more thermal emissions or heat it will produce to achieve the desired effect.1
Light may be:
- absorbed
- reflected (this light will dissipate so quickly it does little to no damage)
- scattered within the tissue (may harm surrounding tissue)
- transmitted within the tissue (may harm surrounding tissue)4,15
Knowing this fact about light will enable you to understand how the below discussed lasers interact with hard and soft tissues in the mouth. The FDA evaluates the effects of each laser on the type of dental tissues it is being used on (i.e., enamel, dentin, pulp, bone or gingivia). They make sure there are no harmful effects from scattered or transmitted light in the mouth.15
Lasers In Dentistry
There are many uses for lasers in the dental field. Depending on the laser in use, one can perform a range of procedures. There is a place for laser use in almost every specialty in the dental field; from dental hygiene to oral surgery. The most common laser procedures are listed below and divided by category.
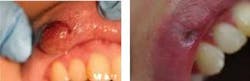
Dental hygienists in some states are using lasers for intrasulcular debridement, in scaling and root planing procedures, aphthous ulcer removal, and pit and fissure sealants. Periodontists are using lasers for osseous surgery and to correct osseous defects (see Figure 1), gingivectomies, frenectomies, gingival curettage, implant placement, and soft tissue crown lengthening. General dentists use lasers for a variety of procedures based on which laser they have purchased. Lasers in the general practice are being used for caries removal, gingival troughing to aid in final impression making, composite curing, to cut on enamel and dentin, dentinal desensitization, crown lengthening, or for the removal of excess gum tissue around healing implant abutments. Lasers are even being used in teeth whitening procedures; laser photons initiate a photochemical activator to increase the response of enamel to hydrogen peroxide.
Lasers also have a place in orthodontics. Lasers — particularly CO2, Nd:YAG, and Er:YAGs — are being used in a range of orthodontic procedures. Instead of acid etching the labial surfaces of teeth before bracket placement, one can laser etch and condition enamel with a laser. Orthodontists can also remove brackets, decrease overall pain with orthodontic adjustments, treat plaque-induced gingivitis, remove plaque-induced hyperplasia of the gums and surgical epulis (see Table 1), and perform fibrotomies (see Table 1).
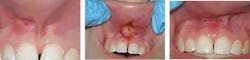
Oral surgeons use lasers for procedures such as biopsies, oral lesion removal (fibromas, papillomas), exposure of an unerupted tooth, and osseoectomy during tooth/root extraction (see Figure 2). Even endodontic procedures are being done with lasers; abscess drainage and pulpotomy as an adjunct to root canal therapy are the most common. Improved tissue healing and decreased postoperative pain by the patient are being reported.20 However, there is concern for the laser's effect on thermal conduction of heat and transmission of energy through dental hard tissue to the external root surface; but a nice advantage is that the laser is replacing the aerosol-producing handpieces in periapical surgeries.20 This can reduce the risk of contamination by blood-borne pathogens.
In 1995, the Food and Drug Administration (FDA) cleared four dental lasers for use on soft tissue only: the argon laser, Holmium:yttrium-aluminum-garnet (Ho:YAG) laser, carbon dioxide laser (CO2), and the Neodymium:yttrium-aluminum-garnet (Nd:YAG) laser.15 In 1997, the Erbium:YAG was the first laser cleared for hard tissue use.14
•Er:YAG: (Erbium doped yttrium aluminum garnet) — This laser is used for the removal of dental decay. It is FDA approved for hard tissue use only. It emits a red beam of light and vaporizes decayed tooth structures, thus replacing the "old" high-speed handpiece and adding to patient comfort by replacing the need for anesthesia in many cases.14 Its wavelength is 2,940nm, which represents the peak absorption of light energy in water.11 This enables the laser to selectively ablate (see Table 1) decay while preserving healthy tooth structure since substances with higher water content are the first to be destroyed. This laser has been shown that it will not increase pulpulal temperature as its cuts.12 For dental hygiene applications, the erbium laser is good for pit and fissure sealants; it will disinfect the preps in case of residual bacteria contamination.
In 1999, Biolase was one of the first companies to market this type of laser.14 Biolase has recently come out with a new laser called the Waterlase, which is FDA approved. It is an Er,Cr:YSGG (Erbium, Chromium doped Yttrium Scandium Gallium Garnet) laser system with a wavelength of 2,780nm. It is showing promise in soft tissue procedures in addition to hard tissue procedures. Procedures such as implant surgery, sulcular debridement, scaling of root surfaces, osseous surgery, and aphthous ulcer treatment are being attempted with the Waterlase. Not only is an Erbium laser indicated for hard tissue treatments, it can be used in soft tissue management and endodontic procedures as well (see Figure 3).
•Argon — Argon lasers have little application to soft tissue management and periodontal therapies. An argon laser is based on the element argon and is used primarily for curing composite resins.15 With the laser, shorter curing times are needed and material strength is stronger when cured with the laser. It emits a blue-green light and has a wavelength of 488nm to 514nm that is the peak wavelength needed for polymerization of the catalyst for curing composites.11 Remember, this laser is located on the visible light spectrum, which is why its wavelength is so much shorter. It is therefore not appropriate for periodontal applications (see wavelength table).
•Ho:YAG: (Holimium doped yttrium aluminum garnet) — A Ho:YAG laser is absorbed by watery tissues and will vaporize, ablate and coagulate soft tissues. Its depth penetration is .4mm or less.15 Its wavelength is 10,900nm and is used on soft tissues and experimentally by oral surgeons on the TMJ.11 For example, it has been used to remove damaged discs separating the condyle of the mandibular bone from the base of the skull among many other types of TMJ surgeries.2 Remember, this laser is located deep into the infrared on the light spectrum. This means the laser will produce lots of thermal emissions, which is why it is not indicated for periodontal procedures.
•Carbon Dioxide: CO2 — Carbon dioxide lasers were the first lasers used in dentistry, thus the most widely studied.20 They emit a wavelength of 10,600nm and with that high of a wavelength, this laser can cut very quickly and deeply. CO2 lasers are mainly used by periodontists and oral surgeons for soft tissue surgeries such as frenectomies, gingivectomies, and biopsies.11 CO2 lasers are highly absorbed in watery tissues and are good at cutting soft tissues without causing bleeding. It is the best laser to use to control bleeding in an area and leaves a residue of carbon, called char, which can serve as a biological dressing, maintaining sterility of surgical sites.15
•Nd:YAG: (neodymium-doped yttrium aluminum garnet) — This laser can penetrate deeper than a carbon dioxide laser and can remove soft tissue just as well. It emits a red beam of light and has a wavelength of 1,064nm in the infrared.11,15 The laser utilizes a free-running (10-6 seconds) pulsed Nd:YAG laser beam and delivers photons in a millionth of a second.1 Free-running pulsed lasers are capable of generating very high peak powers in very short periods of time, which allows safe and rapid ablation of sulcular epithelium in an infected periodontal pocket.
The FDA approved this laser first for sulcular debridement, stating, "The FR Nd:YAG is approved for the removal of diseased or inflamed soft tissue in the periodontal pocket to improve clinical indices including gingival index, gingival bleeding index, probe depth, attachment level and tooth mobility."9
Drs. Robert Gregg and Delwin McCarthy took the concept of the (FR)Nd:YAG one step further and created the now FDA-approved laser-assisted new attachment procedure (LANAP).
The mechanism this laser employs for treating periodontal disease is based on the principle of "bacteria kill."18 This simply means the laser attempts to "kill" the bacteria present in a periodontal pocket. A more technical term for this "killing" is to say the tissue has been ablated.
As hygienists, we know all too well there are many types of bacterial pathogens that can be present in a patient with gum disease. The most common are Porphyromonas gingivalis (Pg), Prevotella intermedia (Pi), Actinobacillus actinomycetemcomitans (Aa) and Eikenella corrodens (Ec).3,18 Dark pigmented and gram-negative anaerobic bacteria are more susceptible to the Nd:YAG laser; as we all know, the above listed bacteria are all gram-negative anaerobic bacteria. This is the simple principle of how this laser "kills" periodontal pathogens. It is designed to remove disease and necrotic tissue selectively from within the periodontal sulcus. Now, you may be wondering how this laser can kill bacteria without affecting surrounding, healthy tooth structures. This laser is not well absorbed in connective tissue and is transparent to water and the cell walls of helper cells such as such as macrophages and endothelial cells. Thus, the "good guys" are left alone and the "bad guys" are eradiated. Healthy tooth structures are left unharmed and the diseased sites of the periodontal pockets are killed.
LANAP not only focuses on killing bacteria in a periodontal pocket, but also on trying to regenerate the attachment apparatus of teeth: the periodontal ligament, cementum, and alveolar bone. It is important to note that this laser is not FDA cleared for all these procedures but may reach that goal someday when more research and longer case studies are available. Let's review quickly the attachment apparatus we are referring to here.
The PDL is the connective tissues that are continuous with the connective tissue fibers of the gingiva and contain collagen fibers that connect it on one side to the cementum and on the other to alveolar bone.3 Fibroblasts are responsible for the fibrous matrix and ground substance of the PDL. They are continually engaged in activities of rebuilding and producing new fibers which are constantly being remodeled. The fibrous matrix is a very important structure and its fibers are inserted into alveolar bone proper and the cementum. There are many different fibers such as dentogingival, dentoperiosteal, transseptal, circumferential, alveolar crest, horizontal, oblique, apical, and interradicular.3 Sharpey's fibers are the terminal fibers of the PDL which insert in alveolar bone and cementum.3
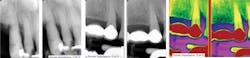
The cementum is a calcified connective tissue that covers the roots of teeth and resembles bone. It is avascular and is the least mineralized of all the calcified tissues of the tooth.3 Alveolar bone has the same biophysical and chemical properties as other bones in the body and is vascular.3 It is made of osteoblasts, osteocytes, osteoclasts, and collagen fibers just to name a few. In periodontal disease, it is these three structures and their subsidies that become greatly affected by the active infection of the mouth. LANAP is showing promising results in regeneration of these important structures, and there is much published research to support this claim (see Figure 4).
Unlike all other lasers on the market, the LANAP laser is unique in its ability to treat periodontal disease. No other laser has clinical trials with results of regrowing PDL, cementum or bone with as much pocket shrinkage. As a hygienist, this laser is the one that perked my interest the most. With the ever changing market of lasers, there may be technology and equipment available in the next couple of years to better treat periodontal disease but to date this laser has shown the most promise.
•Diode laser — The last laser I want to mention is the diode laser. It is not a CO2 laser like many people think it is. Its wavelength varies from 655nm to 980nm.11 At this wavelength, the laser uses heat to "melt" tissue, which is how it cuts or removes tissue. Many practitioners express concern about damages to underlying tissues with the heat this laser emits, but it is good for hemostasis and effective clotting of tissues.20 It can penetrate 2mm to 3mm into tissue and is only indicated for soft tissue applications. This laser is not efficient in deep periodontal pockets because of its design and physical limitations.
The many applications of diode lasers include crown lengthening procedures in cosmetic dentistry and for periodontal management by having a bactericidal effect on periodontal pockets. Oral surgeons use this laser in surgery because of its accurate and bloodless incisions for biopsies, gingivectomies, and frenectomies. It has been used for desensitizing teeth and in teeth whitening procedures in the general dentists' office as well.
The mechanism of this laser in the periodontal pocket is quite simple. First, the electromagnetic energy of the laser beam is absorbed by the carbonized tip. The molecules in the tip are vibrated and converted to heat energy. This makes the tip instantly hotter, above 726 degees centigrade. This heat is then reconverted into electromagnetic energy in the form of incandescence (see Table 1)and the tip then emits visible infrared light. The tip is now referred to as "red hot."1,20 As you can see, much heat is utilized and emitted by this type of laser, and a bad side effect is heat damage to the underlying tissues. This is tissue, of course, that dental hygienists want to preserve because it can give the body something to build back on to regenerate the lost attachment in a deep periodontal pocket. The safety of this laser in the periodontal pocket is under much debate by researchers at this time.
The diode laser is a near-infrared continuous wave (CW) laser, meaning it cannot have the high peak power or microsecond pulse capability as the Free Running (FR)Nd:YAG laser discussed above.20 The pulse duration is much longer at 10-3 than the Fr Nd:YAG's 10-6 duration. The diode in summary will have less peak power and not reach the ablation threshold in soft tissues like the FR Nd:YAG; meaning it will not kill as many pathogens as the Nd:YAG. The diode laser does not produce enough forward emission power per unit of time to cause ablation, and a large portion of the available energy from the CW diode laser is converted to heat, which is also not used for ablation of tissues.20
For example, the diode laser has a surface effect only on the microorganism Porphyromonas gingivalis (Pg), which we all know is a main pathogen in periodontal disease. In a study done by Dr. David Harris at USCF, the Nd:YAG killed 16 times more Pg than the diode.18 For treating moderate to advanced periodontal disease, the diode laser is not nearly as effective in many aspects as the Nd:YAG. By understanding the physical limitations of this laser, you can see how differently it will interact in a periodontal pocket as compared to the Nd:YAG or LANAP laser.
In treating periodontal disease, the diode has not been shown to regenerate cementum, PDL, or bone histologically as some results of the LANAP procedure. There are many hygienists in permitted states using the diode laser in conjunction to scaling and root planing procedures. Research shows that this laser, in conjunction to scaling and root planing, is helpful in eliminating bacteria. Many patients have better bleeding indices and better reduction of pocket depths when this laser is used with scaling and root planing.
The concern I have is that many research articles I have read discuss using the diode laser on patients with 4mm pockets or less or for patients with gingivitis. So, if you have a patient with advanced gum disease with pockets in the 10-12mm range, is this laser really the best tool for treatment? That is the question you need to answer for yourself when you are deciding which type of dental laser fits into your practice needs.
Scope of Practice
Defined under the ADHA scope of practice guidelines for the dental hygienist, only five states allow (with written policy) a hygienist to use lasers to perform soft tissue curettage or periodontal therapy. They are Arizona, California, Colorado, Idaho, and Nevada.
Another 17 states allow laser use but have no written policy on the matter and are Arizona, Delaware, District of Columbia, Indiana, Kentucky, Maine, Michigan, Missouri, Montana, North Dakota, Nebraska, New Mexico, New York, Oklahoma, Oregon, South Dakota, and Washington.
Eleven states do not allow its use with written policy: Alabama, Arkansas, Florida, Georgia, Iowa, Illinois, Ohio, Pennsylvania, Texas, Utah, and West Virginia. Sixteen states are not allowed and have no written policy: Connecticut, Louisiana, Maryland, Massachusetts, Minnesota, Mississippi, North Carolina, New Hampshire, New Jersey, Rhode Island, South Carolina, Tennessee, Vermont, Virginia, Wisconsin, and Wyoming. Hawaii and Kansas are the only states that have not addressed the issue at all and have written policy. The ADHA has a map containing a better view of where to practice with lasers in the United States.
Laser technology is coming and may just revolutionize how we practice dentistry and how we approach periodontal disease. Instead of ultrasonic scalers, we can now offer our patients alternatives. Based on research results that these lasers are producing, it seems we may have stumbled onto more of a "cure" for periodontal disease than just a way of managing it.
If there is a system that can regrow the attachment a patient has lost due to disease instead of just trying to maintain what they have left, then laser technology is an important consideration. Can you imagine a world where we don't have to mechanically remove periodontal pathogens from a patient's sulcus every three months, or cut away bone and tissue with periodontal surgery, or extract teeth just to try to get a handle on their gum disease?
It seems like such a foreign concept to us because it is! In hygiene school, we were taught a certain way to treat patients with periodontal disease, and laser technology teaches us something completely different. We are at the brink of laser use in the dental field and maybe one day we will never have to pick up a noisy, aerosol-producing high-speed drill or ultrasonic again!
For more information regarding laser science and technology, you can refer to the Academy of Laser Dentistry at www.laserdentistry.org or www.adha.org.
Definitions of Terms
•Ablate — The complete removal of material from the surface of an object by erosion vaporization or chipping.
•Atom — The smallest unit to an element, typically indivisible in chemical reactions except for some removal or exchange of particular electrons.
•Fibrotomy — A surgical procedure in which the gingival fibers around a tooth are severed in order to prevent the orthodontically corrected teeth from relapsing.
•Incandescence — Giving off visible light when heated.
•Light Spectrum — The range of all possible electromagnetic radiation. Extends from below the frequencies used for modern radio (at the long-wavelength end) through gamma radiation (at the short-wavelength end). The electromagnetic spectrum of an object is the characteristic distribution of electromagnetic radiation from that object.
•Monochromatic — Consisting of or having one color.
•Photon — A quantum of electromagnetic energy.
•Epulis — A common hyperplastic, fibrous connective tissue lesion. Presents as multiple tissue folds in the anterior alveolar vestibule of firm texture and normal color. Pain is usually experienced at the base of the fold.
•Wavelength — The distance in a periodic wave between two points of corresponding phases.
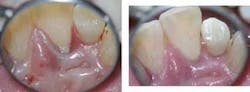
11mm to 12mm pockets all around central and lateral incisors; +3 mobility, note suppuration on photo with circumferential osseous defect on the lingual of #8. At right, post-op of the osseous defect; notice the improved tissue color and contour. Mobility is down to a +1. No probing depths available since you cannot probe for nine months post-op. Photos courtesy of Donato Dental.
About the Author
Lisa Dowst-Mayo received a bachelor's degree in dental hygiene from Baylor College of Dentistry in 2002. She is a member of ADHA, TDHA and is the president-elect of the Dallas Dental Hygiene Society. She works as a full-time dental hygienist in private practice in Flower Mound, Texas, at Cross Timbers Dental. She will be teaching a continuing education class at the annual Southwest Dental Conference in Dallas on laser dentistry in 2009.
References
1. Bornstein E. Method and Dosimetry for Thermolysis and Removal of Biofilm in the Periodontal Pocket With Near-Infrared Diode Lasers. Dentistry Today, April 2005; 24(4): 1-9.
2. Butler S. Laser-Assisted Temporomandibular Joint Surgery. Lasers in Maxillofacial Surgery and Dentistry. Thieme, 1997; p.151-162.
3. Darby M. Mosby's Comprehensive Review of Dental Hygiene Fourth Edition. Mosby's, 1998; p. 44-49.
4. Dean B. Using the Er,Cr:YAGG Laser. Concepts in Laser Periodontal Therapy, 2005.
5. Gregg R, McCarthy D. Eight Year Retrospective Review of Laser Periodontal Therapy in Private Practice. Dentistry Today, Feb. 2003; 22(2): 1-4.
6. Gregg R, McCarthy D. Laser ENAP for Periodontal Bone Regeneration. Dentistry Today, May 1998; 17(5): 1-3.
7. Gregg R, McCarthy D. Laser ENAP for Periodontal Ligament (PDL) Regeneration. Dentistry Today, Nov 1998; 17(11): 1-3.
8. Gregg R, McCarthy D. Laser Periodontal Therapy for Bone Regeneration. Dentistry Today, May 2002; 21(5): 1-4.
9. Harris D, Gregg R, McCarthy D, Colby L, Titl L. Laser-assisted new attachment procedure in private practice. General Dentistry, Sept-Oct 2004; 52(5).
10. Harris D, Gregg R, McCarthy D, Colby L, Titl L. Sulcular Debridement with Pulsed Nd:YAG. Lasers in Dentistry VIII, Proceedings of SPIUE Vol. 4610 (2002). Available at http://www.millenniumdental.com/research/jan-02.html. Accessed Sept 26, 2007.
11. Institute for Laser Dentistry. Laser Wavelength. Institute for Laser Dentistry, 2000-2002. Available at http://www.laserdentistry.ca/faq/laser_wave.html. Accessed Sept 26, 2007.
12. Jesse J, Desai S, Oshita P. Ruby to YSGG. The Evolution of Lasers in Dentistry, 2005.
13. Keanna G. Biological Dentistry. New Health Insight, A New Frontier in Healthcare, Jan-Feb 2006. Available at www.newhealthinsight.com/index.php/Biological-Dentistry/Non-Surgical-Laser-Gum-Treatment.html. Accessed Sept 23, 2007.
14. Kurtzweil P. Dental More Gentle with Painless ‘Drillings' and Matching Fillings. FDA Consumer Magazine, May-June 1999. Available at http://fda.gov/fdac/features/1999/399_dent.html. Accessed Nov 16, 2007.
15. Lewis R. Lasers in Dentistry. FDA Consumer Magazine, Jan-Feb 1995. Available at www.fda.gov/bbs/topics/CONSUMER/CON00296d.html. Accessed Sept 23, 2007.
16. Lowe R. Dental Products Report. Thomson Healthcare/Dental Products Report, May 2002. Available at http://www.biolase.com.mx/clinicalarticles/Lowe_Cosmetic_DPR-5-02.pdf. Accessed Jan, 2008.
17. Hafez MI, Coombs RRH, Zhou S, McCarthy ID. Journal of Clinical Laser Medicine & Surgery. October 1, 2002, 20(5): 251-255.
18. Neill M, Bader H. Clinical Efficacy of the Nd:YAG Laser for Combination Periodontitis Therapy. Practical Periodontics and Aesthetic Dentistry, August 1997; 9(6):1-9.
19. Sanderson J. Dental Laser Basics. Dental Health Network, 1997-2007. Available at www.dental--health.com/dental_laserbasics.html. Accessed Sept 26, 2007.
20. Todea C. Laser Applications in Conservative Dentistry. TMJ, 2004; 54(4): 392-405.
21. Yukna R, Carr R, Evans G. Histologic Evaluation of an Nd:YAG Laser-Assisted New Attachment Procedure in Humans. Internal Journal of Periodontics & Restorative Dentistry, Nov. 2007; Vol.27: 577-587.