Pharmacology 101
Knowledge of drug classes can help hygienists improve the safety and quality of dental hygiene care.
by Howard M. Notgarnie, RDH, MA
Many of the prescribed drugs used today are responses to conditions related to lifestyle. Nowadays, people frequently use drugs for pain, inflammation, emotion, and improvements to life. Although the use of drugs is not frivolous for these problems, people would be better off preventing their need or seeking intervention earlier in the disease process. Furthermore, each class of drugs for these problems shows multiple repetitions. Knowledge of these classes of drugs improves the safety and quality of dental hygiene care by giving the dental hygienist the opportunity to avoid or decrease the severity of adverse reactions. This information can also open up the conversation about the drugs, causes of disease those drugs treat, and sources of additional care that may help prevent, control, or eliminate those diseases.
Cytochrome P450
Many of the drugs discussed in this article undergo transformation by an important group of enzymes known collectively as cytochrome P450. Kalra describes cytochrome P450 as a group of isoenzymes, each of which can break down multiple endogenous and exogenous chemicals by combining the chemical with molecular oxygen in the presence of NADPH (hydrogenated nicotinamide adenine dinucleotide phosphate) to make an oxygenated form of the chemical plus water. The site of this enzyme reaction in the cell is primarily on endoplasmic reticulum. The oxidized form of the chemical is water-soluble rather than lipid soluble, as in its original form, therefore more amenable to elimination in urine or feces. Most cytochrome P450 is in the liver, but it is also present in many other organs. Its name comes from its effect as a pigment, absorbing light at 450 nm when bound to carbon monoxide. The nomenclature for this group consists of “CYP” followed by a number, a letter, and another number. The first number represents the family, enzymes that contain at least 40 percent of the amino acid sequence in common. The letter represents the subfamily, enzymes in the same family that have at least 55 percent of the amino acids in common. The second number represents the gene, of which humans have at least 57 for cytochrome P450.
Most drug interactions concerning cytochrome P450 involve inhibition due to competition for the binding site of the enzyme. CYP 1, 2, and 3 are responsible for 94 percent of drugs metabolized in the liver. Less than 30 years ago, the only known form of cytochrome P450 was isolated, but we now know there is a wide range of genetic variability for cytochrome P450 leading to variation in effects of drugs within the population due to the amount and effectiveness of the cytochrome P450 isoenzymes produced. People who metabolize drugs slowly will experience higher toxicity of ingested drugs, while those who have more effective or greater number of isoenzymes may break the drugs down before the drugs have a chance to take effect. Likewise, medications taken as prodrugs that are metabolized to their activated forms may have insufficient effect for slow metabolizers and cause adverse reactions for fast metabolizers. Because of this knowledge, researchers are looking forward to tailoring prescriptions to cytochrome P450 genotype or phenotype by altering drug choice, dosage, or a competitive inhibitor of the enzyme.
A common reason for drug interactions is that the same enzyme metabolizes both drugs involved in the interaction. Competition for the same site on the enzyme results in one or both of the drugs maintaining higher blood concentrations than intended. Adverse reactions follow. Another well-known effect is enzyme induction. In this case, the drug causes an increase in the enzyme that metabolizes it. One mechanism of enzyme induction is through gene activation. The drug binds to a carrier protein in the cytosol. A nuclear translocater then transports the drug-protein complex through the nucleus to a receptor that activates DNA transcription. Barbiturates and many antibiotics induce CYP3A this way. Two other mechanisms of enzyme induction are stabilization, so the enzyme does not degrade as quickly, and increased translation of RNA to protein. These two mechanisms are the cause of ethanol and isoniazid inducing CYP2E1. The increased level of enzymes due to induction leads to quicker elimination, reduced blood levels, and tolerance to the drugs metabolized by the induced enzyme. Cytochrome P450 levels top at up to two weeks after peak levels of the inducer molecule, and the enzyme’s normal levels return up to three weeks after the inducer is eliminated. Additional inducers include rifampicin, glucocorticoids, macrolides, anticonvulsants, polychlorinated biphenyls, St. John’s wort, and polycyclic aromatic hydrocarbons (examples: tobacco and charcoal).
Inhibition is typically specific to the isoenzyme. Reversible inhibition occurs when drugs are competing for the binding site. For cytochrome P450, this is at the heme moiety, where the molecule undergoes oxidation. Cimetidine, an H2 receptor antagonist, also competitively inhibits CYP3A4 and 2D6 weakly, while ketoconazole is a stronger inhibitor due to its more lipophilic character. Irreversible inhibition, for example, erythromycin occurs when the oxidized form of the drug has a strong affinity for the binding site, forming a complex that permanently blocks the functional unit of the enzyme. Additional cytochrome P450 inhibitors include protease inhibitors used to combat HIV, selective serotonin reuptake inhibitors (SSRIs), and antifungal agents.1
Nonsteroidal Anti-inflammatory Drugs
Rayyan, Williams, and Rigas identify Edmund Stone as the first person to isolate a salicylate from white willow bark in 1829, although the bark’s anti-inflammatory effects were known for thousands of years prior to that.2 There are numerous nonsteroidal anti-inflammatory drugs (NSAIDs). The mechanism of their action is inhibition of cyclooxygenase enzymes cox-1 and cox-2. By reducing these enzymes’ function, the body produces less of the prostaglandins involved in the inflammation that causes pain. The cox-1 enzyme is also important for platelet production, thus its inhibition reduces the ability to clot. Inhibition of cox-1 also reduces the ability of the stomach lining to protect from acids. NSAIDs selective for cox-2, such as celecoxib, rofecoxib, and valdecoxib, limit these side effects because the cox-2 enzyme is not located in the stomach.3
There are additional potential uses for this class of drug. Research teams in Mexico recently found NSAIDs reverse ethanol-induced oxidation of lipids.4 People with some variants of the gene for cox-2 have an increased risk of basal cell carcinoma. Researchers are studying the possibility that NSAIDs may reduce this risk, although so far, the benefit of NSAIDs for this purpose seems small.5 A review of 1,790 prospective studies on the protective effects of NSAIDs against colorectal cancer and colonic adenoma found significant reduction in the risk of these conditions. However, the reduction in risk is not enough to justify the increased risk of gastrointestinal and cardiovascular side effects in people not at high risk of these neoplasms. Cox-2 inhibitors increase the risk of myocardial infarction, hypertension, and renal toxicity. NSAIDs increase the risk of ulcers and hemorrhage in the gastrointestinal tract, especially for those who are already at risk of these conditions. These risks can be ameliorated by using cox-2 NSAIDs or by adding histamine-2 (H2) receptor antagonists or proton pump inhibitors.6
The first recognition of NSAIDs as a possible treatment or prevention of cancer was in 1973, when indomethicin reduced the size of fibrosarcoma in mice. Epidemiological studies confirmed the protective effect of NSAIDs. The benefit is due to effects of NSAIDs on cell kinetics. Mutations in several genes involved in tumor growth, tumor suppression, and DNA repair led to excess growth of mutated cells vs. death of those cells. NSAIDs reduce cell proliferation and increase cell death. These effects are directly on cells as well as indirectly through their effects on prostaglandin production as well as the enzymes PPARd (peroxisome proliferator activated receptor delta) and NF-¿B (Nuclear Factor kappa B). Further, the drugs inhibit blood vessel formation, thereby reducing the nutrient supply for the tumors. The direct effect on cells is to alter the amounts of cyclins and cyclin-dependent kinases involved in mitosis. The indirect effect, inhibition of cyclooxygenase during its conversion of arachidonic acid to PGG2 (prostaglandin G2), is believed to be due to prostaglandins’ stimulation of tumor cell growth. Further, endogenous chemicals promoting growth and tumors induce cox-2.
Cyclooxygenase also activates some carcinogens through its peroxidase effects. PPARd is part of a complex that activates transcription upon binding to DNA. The NSAID sulindac prevents this binding. NSAIDs inhibit vascular formation by disrupting mitosis at the stage of nuclear translocation. Another possible mechanism of NSAIDs may be an increase in an immune product, MHC class II, a molecule that presents antigens on macrophages and B cells. Cells with poorly functioning DNA repair mechanisms are more susceptible to self-destruction in the presence of NSAIDs. NF-¿B, which activates transcription of several genes, is inhibited from getting to the nucleus due to the NSAID’s effects on its precursor failing to break down to the NF-¿B component.
Gastrointestinal and renal side effects of NSAIDs are common. Hypersensitivity and intoxication to the drugs are less common; however, in 1997 alone, there were 16,500 deaths related to their use.2
Statins
Statins occur naturally in several species of fungus.7 Like NSAIDs, statins can reduce clotting8 and inflammation.9 However, they are prescribed primarily to normalize blood cholesterol and lipid levels through their inhibition of the enzyme 3-hydroxy-3-methyl-glutaryl coenzyme A reductase (HMG coA reductase).10 This enzyme is one of the steps in the process of forming cholesterol where mevalonate is formed.11 Statins also benefit patients with “aortic valve stenosis, diabetes mellitus, sepsis, and heart failure” regardless of the effect on blood concentrations of cholesterol.12 A preliminary study shows atorvastatin not only reducing cholesterol levels, but reducing the sympathetic output of the autonomic nervous system to a more balanced level with the parasympathetic output.10 Excessive amounts of cholesterol can inhibit mevalonate kinase, important in cholesterol production, and cobalamin adenosyltransferase, which produces a chemical needed for mitochondrial function. Statins increase the function of the above two enzymes.13 Lovastatin9 and simvastatin protect nerve cells from toxicity by increasing “expression levels of a gene encoding for a major cell survival protein, bcl-2”14 found in the membranes of several cell organelles.
Statins can decrease the formation of new blood vessels. Lovastatin was used in an in vitro study that might lead to treatment of endometriosis. In this study, lovastatin decreased angiogenesis and growth of endometrial tissue in vitro.15 There is some growing evidence that this effect is due to suppression of NF-¿B, which becomes more active upon inflammation, growth, and “oxidative stress,” thereby inducing transcription in proliferating cells.16
Prior to surgery, beta (ß) blockers and possibly statins and alpha2 (a2) agonists are helpful in reducing the risk of postoperative myocardial infarction, angina, and death due to cardiac complications. Statins in particular help by reducing thrombosis and proliferation of atheromata (the lesions characteristic of atherosclerosis) and by preventing white blood cells from adhering.17 Statins stabilize atherosclerotic plaque and reduce free radicals and inflammation in endothelium.11
Another enzyme important in controlling cholesterol is cholesterol esterase transfer protein (CETP). CETP is attached to high density lipoproteins (HDL) for the purpose of transferring cholesterol from HDL to low density lipoproteins (LDL). Normally this is beneficial for the proper metabolism of cholesterol, but the high levels of cholesterol and LDL in most Americans’ blood make the function of CETP risky. Although drugs under study that inhibit CETP raise HDL concentrations, a recent study found an unsafe increase in blood pressure associated with the drugs’ use.18
Recent studies are showing that even people with low cardiac risk can have the risk of coronary heart disease decreased with statin drugs due to their ability to lower LDL blood concentrations and reduce the size of atherosclerotic lesions. Currently the goal is to have LDL less than 100 mg/dL for high-risk patients with some experts leaning to 70 mg/dL, while people with low risk of coronary heart disease face a goal of less than 160 mg/dL. There are, however, risks in using statins due to toxic effects on the liver and skeletal muscles. Blood levels of liver transaminase and creatine kinase are sometimes elevated, although distinct liver damage is rare. Adverse reactions including muscle destruction are more likely at higher doses or when statins are combined with other drugs that are metabolized by CYP3A4. A potential for reducing these drug interactions is evident in rosuvastatin, which has alternate isoenzyme pathways, 2C9 and 2C19. Muscle destruction increases blood levels of myoglobin, which can lead to kidney failure.19
Consumption of grapefruit affects the metabolism of drugs by reducing the amount of CYP3A4 in the intestine and possibly by reducing the absorption of those drugs. The overall effect is to increase the blood concentrations of drugs metabolized by this isoenzyme, which include, along with statins, many antiarrhythmics, calcium channel blockers, immunosuppressants, ACE inhibitors, and protease inhibitors. The specific components of grapefruit that cause this are unknown. The intestinal isoenzyme may be at lower levels after grapefruit consumption for one to three days. Current recommendations are to avoid grapefruit for 72 hours prior to use of those drugs.20 Phenytoin, on the other hand, a drug used to control seizures induces the isoenzyme CYP3A4,21 as do glucocorticoids, carbamazepine, and phenobarbitol, thereby reducing the effectiveness of statins.1
Selective Serotonin Reuptake Inhibitors
Selective serotonin reuptake inhibitors (SSRI) are used for their antidepressant effects, but they have a multifaceted effect on physiology that may be beneficial for cardiovascular health. A review of the literature on treatment of depression in patients with acute cardiac syndromes (ACS) suggests an equivocal relationship between treatment of depression and prognosis for ACS. Treatment of depression included varying roles of SSRIs, diet, and behavioral and cognitive psychological care. SSRIs may reduce platelet activity and sympathetic nervous function as well as improve immune system function. This is particularly important because people hospitalized for ACS (such as angina and myocardial infarction) frequently develop depression, which worsens the prognosis of these patients. The attraction of SSRIs to the serotonin transporter protein reduces the amount of serotonin in platelets, thus reducing their ability to aggregate.
The two primary areas where the body produces serotonin are the raphe nuclei of the brain stem and the enterochromaffin cells found in the gastrointestinal epithelium. The body stores much of the serotonin in platelets after it is released from these structures. The benefits of SSRIs over tricyclic antidepressants are fewer side effects on a1 and a2 histamine, muscarine, and dopamine receptors. Tricyclic antidepressants’ effects on these receptors may cause hypotension and arrhythmias where SSRIs do not. SSRIs, however, do have drug interactions with ß blockers, ACE inhibitors, statins, antiarrhythmics, calcium channel blockers, and warfarin due to their inhibition of at least six isoenzymes of cytochrome P450.
SSRIs seem to enhance immune function by increasing valuable cytokines. The improvements in the immune system may be a factor in the reduced symptoms of depression and heart disease. Although the mechanisms are still under study, a promising hypothesis of the effects of SSRIs on cardiac prognosis is that while acute coronary syndromes cause overload of sympathetic nervous activity, SSRIs reduce sympathetic nervous activity, balancing the autonomic nervous system, thus increasing heart rate variability (HRV), which is important for heart function. Depression reduces HRV as well as the body’s reflex to change blood pressure in response to need. Furthermore, depression increases platelet aggregation apparently due to increased levels of platelet factor 4 and ß thromboglobulin.
There are a number of serotonin receptors, which have varying effects on their target organs. However, most studies are indicating that SSRI-induced vasodilation is not due to serotonin receptors on blood vessel endothelium, but due to effects on calcium channels.12
Although SSRIs have widely varying affinity for the serotonin transporter, none of them binds at greater than 90 percent of transporter enzymes present, regardless of dosage. While this is sufficient for many cases of depression, a higher binding rate needed for obsessive-compulsive disorder is available with the use of clomipramine.22
SSRIs enhance the effectiveness of cognitive behavioral therapy (CBT), in contrast with benzodiazepines, which diminish the effectiveness of CBT, and tricyclic antidepressants, which have mixed effects on CBT. Wright suggests this benefit of SSRI is due to improvements in “learning and memory.” An interesting prospect in combined usage of behavioral and pharmacological therapy is that the two types of treatment work in opposite directions with respect to the evolutionary development of the brain. Drugs have their direct effect on the more primitive part of the brain, leading to the opportunity for cognitive changes. Cognitive therapy, on the other hand, has its direct effect on cortical structures of the brain, which then leads to effects on primitive structures.23
The Harvard Mental Health Letter indicates that in some cases there may be important interactions among antidepressants, genetic susceptibility, and folic acid. In particular, people who have a gene that keeps them from using folic acid as effectively as they should have a higher risk of mental disorders, including depression. Adding a folic acid supplement to the diet of people on an SSRI showed improvement in the effectiveness of the SSRI in some people. Furthermore, folic acid can reduce the blood levels of homocysteine, an amino acid associated with depression.24 Nursing Standard reports that elderly people taking SSRIs have an increased risk of falling and fracture due to osteoporosis.25
Gastric Acid Inhibitors and Related Drugs
Proton pump inhibitors have become the most popular prescription for relief of gastroesophageal reflux disease, but they are expensive, especially when used chronically. Furthermore, H2 receptor antagonists raise gastric pH much faster and are very effective for use on an as needed basis in nonerosive esophagitis.26 Proton pump inhibitors can be effective in the treatment of ulcers and reflux in the gastrointestinal tract. A recent meta-analysis of studies indicates use of this class of drugs in people who have had treatment for ulcers reduces bleeding and the need for further operative treatment. In Asians, the medications also are associated with reduction in all-cause mortality. One particular factor related to the pharmacological aspects of the drugs is that Asians are more likely to be slow metabolizers of this class of drug. The primary benefit of these drugs is that when they inhibit gastric acid production, the increased pH supports clot formation and stabilization so that ulcers can heal.27
Durner and Rea explain the role of stress in forming gastric ulcers. Stress induces parietal cells to produce more acid than they normally would. At the same time, there may be insufficient blood flow to the stomach due to cytokine production; the mucous membrane will produce insufficient glycoprotein and bicarbonate to protect itself from acidic tissue damage.28
Parsons and Ganellin indicate histamine was first identified in 1910, and the first useful antihistamines, based on aniline derivatives, became available for use in humans in 1942. Mast cells store histamine. The enzyme histidine decarboxylase forms histamine from 1-histidine. Both diamine oxidase and histamine methyl transferase can break down histamine. Histamine reaches receptors, inducing increased heart rate, vasodilation, and gastric secretion while decreasing uterine contractions. For gastric H2 histamine receptors, histamine acts as a messenger molecule that leads to adenylate cyclase activation to form cyclic AMP, which acts as a messenger inside the cell.
Antihistamines used to control vasodilation at H1 receptors do not have an effect on gastric acid secretion because there is a distinctly different receptor, H2, responding to histamine. Furthermore, there is an H3 receptor on nerves. Researchers developed H2 antagonists by producing chemical analogues to histamine. Cimetidine was the first compound of this class that was not excessively toxic. These are particularly effective because H2 receptor antagonism reduces gastric acid even in the presence of other stimulators of acid production: gastrin, acetylcholine, vagus nerve stimulation, and food ingestion. Ranitidine is stronger than cimetidine and does not affect cytochrome P450 the way cimetidine does.
Many H1 receptor antagonists are able to pass through the blood-brain barrier, affecting neurons causing sedation, but the brain also has H3 receptors, whose activation decreases cyclic AMP production and inhibits production and discharge of histamine. H4 receptors are much more similar in composition to H3 receptors than to H1 or H2 and appear on immune cells for the activation of eosinophils, basophils, mast, T, and dendritic cells. Prospective uses for H3 and H4 antagonists include cognitive disorders, sleep disorders, epilepsy, inflammatory diseases, and obesity.29
H2 receptor antagonists are associated with greatly reduced metastatic lesions in cancer patients. Although the mechanism is unknown, most theories involve the drugs’ competing with histamine’s suppression of tumor infiltrating lymphocytes. T lymphocytes and natural killer cells not suppressed by histamine seem better able to infiltrate tumors and produce cytotoxins that destroy cells through fas-induced and perforin-induced pathways. H2 receptor antagonists may also have a direct effect slowing growth of cancer cells, some of which also have H2 receptors.30
Histamine is present in keratinocytes, which have H1 and H2 receptors. While the mechanism is unknown, H1 and H2 receptor antagonists applied locally increase healing of the skin’s barrier function. Histamine in the skin increases upon exposure to ultraviolet light and enhances the effect of the inflammatory cytokine, interleukin 6.31
Histamine appears to have a self-regulatory effect. High concentrations induce basophils to absorb histidine through its agonist effect on H1 receptors.32
Steroids
In addition to antihistamines, corticosteroids are common for relief of allergies and other causes of inflammation. Long-term use of steroids may be necessary in some cases, but the side effects do pose significant risk. A pilot study for the treatment of autoimmune uveitis points out that the suppressed immune system puts patients on these drugs at higher risk of infections and neoplasms. In response to this problem, they tested the benefit of using echinacea (an herb known to modulate the immune system and reduce inflammation) along with standard steroid therapy and found that the herb does indeed reduce the amount of time patients with autoimmune uveitis need treatment with steroids.33
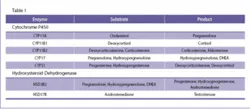
Adrenocortical and gonadotropic hormones are derived from cholesterol. Two major enzyme groups are responsible for hydroxylation, oxidation, and dehydrogenation of cholesterol and its derivatives, hydroxysteroid dehydrogenase (HSD) and cytochrome P450. In the adrenal cortex, we see steroids produced via the following enzyme pathways shown in Table 1.
The presence of these steroids is important in sexual differentiation.34 Their hormonal functions occur by activating transcription in cell nuclei. To increase muscle mass and strength, competitive athletes frequently use androgenic steroids; however, the chronic use of steroids causes risk to multiple organs and systems. Hypertrophy and scarring of the heart’s left ventricle leads to impaired blood flow. Steroids affect the “vascular response to norepinephrine,” which can cause lethal arrhythmias. Their effects of dyslipidemia and increased platelet aggregation cause atherosclerosis and increase the risk of thrombosis. Steroids increase the risk of abnormal function and cancer of the liver. Steroids have a deleterious effect on collagen composition, making tendons more likely to tear upon exertion. They lead to aggressive and manic behavior, and their withdrawal leads to depression. Excess anabolic steroids lead to deleterious changes in the reproductive system, some of which are irreversible. Steroids affect skin due to sebaceous gland hypertrophy. Chronic users show excess facial hair, male pattern baldness, stretch marks, and acne. Steroids increase hemoglobin levels and risk of infection. Larger muscles due to steroid use require greater oxygen intake and CO2 exhalation. Steroids induce cytochrome P450, thereby increasing metabolism of other drugs, including anesthetics.35
Dehydroepiandrosterone (DHEA) is available over the counter, marketed for its anabolic effects. It helps women taking glucocorticoids for systemic lupus erythematosus from losing bone density. People produce DHEA mostly in the adrenal cortex, but in several other organs as well. It is “the most abundant endogenous adrenal steroid,” and most of it is in the inactive, sulfate form until converted in tissues throughout the body. DHEA is the precursor to half the androgens in men and most of the estrogens in women. Its highest concentration is in the brain, where it acts on “neurotransmitter receptors (gamma-aminobutyric acid-benzodiazepine receptor complex and N-methyl-D-aspartate).” DHEA may have an effect on T cell production of interferon and several interleukins, based on in vitro studies. DHEA is saved at the kidneys through tubular reabsorption. A possible reason for the bone-sparing effect of DHEA is its conversion to estrone, although the anabolic effects of DHEA are still under investigation. Use of exogenous DHEA can cause acne, hirsutism, blood in the urine, and elevated creatinine blood concentrations. There is some evidence that DHEA can alter lipid and cholesterol levels as well as the complement system important in immune response. It reduces the blood levels of adrenocorticotropic hormone. Drugs that increase CYP3A4 will decrease DHEA, and DHEA will competitively inhibit other drugs catabolized by that isoenzyme. DHEA is an antagonist to 5-a-reductase inhibitors (which treat benign prostatic hypertrophy). DHEA inhibits platelet aggregation. Insulin reduces DHEA levels for unknown reasons. Interactions may also occur with gonadotropic hormones and drugs used to regulate them.36
Vitamin D (calciferols)
This vitamin is actually a steroid hormone. The hydroxylation of 25-hydroxycholecalciferol to the active form of the vitamin, 1,25-dihydroxycholecalciferol occurs mostly in the kidneys by CYP27B1. The production of the active form of vitamin D begins the mechanism of regulating blood concentration of calcium. Furthermore, blood concentrations of 25-hydroxycholecalciferol are associated with lower risk of chronic disease. The skin can photosynthesize vitamin D from 7-dehydrocholesterol in the form of cholecalciferol. That and ergocalciferol from plants are synthesized to the main storage form, 25-hydroxycholecalciferol (calcidiol) in the liver by CYP27A1. The liver also converts this to 24,25-dihydroxycholecalciferol with CYP24. CYP27B1 also exists to a lesser degree in and near tissues and cells that may use vitamin D in ways we are just beginning to recognize. The isoenzyme is present in colon, muscle, and immune cells. The vitamin has a specific carrier protein in plasma. Insufficient vitamin D causes low absorption and blood concentrations of calcium and increased parathyroid hormone levels. In short, lack of sunlight or ingested vitamin D causes poor bone metabolism. However, it is also becoming clear that chronic diseases are more likely in people with deficiency of vitamin D. Insufficient vitamin D is associated with metabolic syndrome, a collection of poor health conditions that lead to a high risk of cardiovascular disease: obesity, dyslipidemia, hypertension, insulin resistance, increased inflammatory response, and increased clotting response. Currently it is unknown whether the relationship between vitamin D concentration and metabolic syndrome is causal; however, there are some distinct metabolic relationships. Excess body fat is highly associated with reduced 25-hydroxycholecalciferol levels and with hyperparathyroidism. The parathyroid hormone stimulates CYP27B1 to convert more of the provitamin to the active 1,25-dihydroxycholecalciferol in order to increase blood calcium by reabsorption in the kidneys, and absorption through the intestines and resorption from bone. Vitamin D is important in producing sufficient amounts of insulin in response to glucose and arginine, apparently by supporting ß cell production of insulin in the pancreas. Vitamin D may also be involved in regulating the renin-angiotensin system, and thus blood pressure. 1,25-dihydroxycholecalciferol has receptors on monocytes and T cells, probably accounting for the inhibition of several interleukins and tumor necrosis factor-a, but how this is related to metabolic syndrome is unknown.37
Vitamin D activates or inhibits transcription, depending on the target gene, at the nucleus by reaching vitamin D receptor with a ligand. Hydroxylation of calciferols to active forms occurs in liver, kidneys, and the small intestine through various isoenzymes of cytochrome P450. Parathyroid hormone increases CYP27B and decreases CYP24 to increase 1,25-dihydroxycholecalciferol, while the vitamin in that form, through a vitamin D receptor, causes the opposite for those two enzymes. Phenobarbital, phenytoin, and carbamazepine, used long term, cause osteomalacia in a manner mimicking vitamin D deficiency. This is thought to be due to excessive catabolism of 1,25-dihydroxycholecalciferol. These drugs activate steroid and xenobiotic receptor (SXR, a nuclear receptor) in the liver and small intestine. Activation of SXR regulates several cytochromes and other enzymes to control the metabolism of foreign material. Apparently, this effect is by SXR inhibiting vitamin D receptor’s tendency to increase CYP24, rather than SXR having a direct effect on CYP24. The authors refer to this effect as “crosstalk” between the receptors, which is also a known phenomenon between SXR and an androstane receptor.38
Conclusion
When we understand the mechanisms of a drug’s action and metabolism, we are in a much better position to care for our clients effectively. (See the accompanying sidebar for a summary of drug characteristics.) This information provides us a basis of concern when working with a client taking multiple medications, of drug choice when providing clinical care, and of decisions when choosing information with which to educate clients. Understanding the effects of drugs provides dental hygienists the serendipity to identify potential conditions for which a client may need referral to another health-care practitioner and to take an authoritative approach toward our part of a client’s interdisciplinary care.
Knowledge of risks of these drugs also should be a warning to ourselves to identify lifestyle habits that we can improve in ourselves and encourage in our clients and others we know. Maintaining health without drugs through diet, exercise, and other lifestyle behaviors greatly improves one’s chance of continuing a healthy life. That should be a general goal of preventive health professionals like dental hygienists. Click here to view Summary of Drug Characteristics
About the Author
Howard M. Notgarnie, RDH, MA, practices dental hygiene in Colorado, and has eight years’ experience in official positions in dental hygiene associations at the state and local levels.
1. Kalra BS. Cytochrome P450 enzyme isoforms and their therapeutic implications: An update. Indian Journal of Medical Sciences 2007; 61(1):102-116.
2. Rayyan Y, Williams J, Rigas B. The role of NSAIDs in the prevention of colon cancer. Cancer Investigation 2002; 20(7/8):1002-1011.
3. Ogbru O. (2005). Nonsteroidal antiinflammatory drugs (NSAIDs). Retrieved March 26, 2006, on http://www.medicinenet.com/nonsteroidal_antiinflammatory_drugs/article.htm.
4. Zentilla de Piña M, Sandoval-Montiel A, Serrano-Alessandri L, Montalvo-Jave E, Zentella-Dehesa A, Piña E. Ethanol-mediated oxidative changes in blood lipids and proteins are reversed by aspirin-like drugs. Archives of Medical Research 2007; 38(3):269-275.
5. Vogel U, Christensen J, Wallin H, Friis S, Nexo BA, Tjonneland A. Polymorphisms in COX-2, NSAID use and risk of basal cell carcinoma in a prospective study of Danes. Mutation Research: Fundamental & Molecular Mechanisms of Mutagenesis 2007; 617(1/2):138-146.
6. Rostom A, Dubeé C, Lewin G, Tsertsvadse A, Barrowman N, Code C, Sampson M, Moher D. Nonsteroidal anti-inflammatory drugs and cyclooxygenase-2 inhibitors for primary prevention of colorectal cancer: A systematic review prepared for the U.S. Preventive Services task force. Annals of Internal Medicine 2007; 146(5):376-396.
7. Seidl M. Industrial uses of fungi. The Environmental Reporter 2006; 4(9). Retrieved April 14, 2007, on http://www.emlab.com/s/sampling/env-report-09-2006.html.
8. Schäfer A, Fraccarollo D, Vogt C, Flierl U, Hemberger M, Tas P, Ertl G, Bauersachs J. Improved endothelial function and reduced platelet activation by chronic HMG-CoA-reductase inhibition with rosuvastatin in rats with streptozotocin-induced diabetes mellitus. Biochemical Pharmacology 2007; 73(9):1367-1375.
9. Sarkey JP, Richards MP, Stubbs EB. Lovastatin attenuates nerve injury in an animal model of Guillain–Barré syndrome. Journal of Neurochemistry 2007; 100(5):265-1277.
10. Szramka M, Harriss L, Ninnio D, Windebank E, Brack J, Skiba M, Krum H. The effect of rapid lipid lowering with atorvastatin on autonomic parameters in patients with coronary artery disease. International Journal of Cardiology 2007; 117(2):287-291.
11. Bonovas S, Sitaras NM. Does pravastatin promote cancer in elderly patients: A meta-analysis. Canadian Medical Association Journal 2007; 176(5):649-654.
12. Van Melle JP, De Junge P, Van Den Berg MP, Pot HJ, Van Veldhuisen DJ. Treatment of depression in acute coronary syndromes with selective serotonin reuptake inhibitors. Drugs 2006; 66(16):2095-2017.
13. Murphy C, Murray AM, Meany S, Gåfvels M. Regulation by SREBP-2 defines a potential link between isoprenoid and adenosylcobalamin metabolism. Biochemical and Biophysical Research Communications 2007; 355(2):359-364.
14. Johnson-Anuna LN, Eckert GP, Franke C, Igbavboa U, Müeller WE, Wood WG. Simvastatin protects neurons from cytotoxicity by up-regulating Bcl-2 mRNA and protein. Journal of Neurochemistry 2007; 101(1):77-86.
15. Esfandiari N, Khazaei M, Ai J, Bielecki R, Gotlieb L, Ryan E, Casper RF. Effect of a statin on an in vitro model of endometriosis. Fertility and Sterility 2007; 87(2):257-262
16. Guo Sun-Wei. Nuclear Factor-κB (NF-κB): An unsuspected major culprit in the pathogenesis of endometriosis that is still at large? Gynecologic and Obstetric Investigation 2007; 63(2):71-97.
17. Flood C, Fleisher LA. Preparation of the cardiac patient for noncardiac surgery. American Family Physician 2007; 75(5):656-665.
18. Anonymous. New drug fizzles at raising HDL. Harvard Heart Letter 2007; 15(7):1-2.
19. Jacobson T. The safety of aggressive statin therapy: how much can low-density lipoprotein cholesterol be lowered? Mayo Clinic Proceedings 2006; 81(9):1225-1231.
20. Stump AL, Mayo T, Blum A. Management of grapefruit-drug interactions. American Family Physician 2006; 74(4):605-608.
21. Trifiro G. Drug-drug interactions and statin therapy. Southern Medical Journal 2006; 99(12):1325-1326.
22. Zipursky RB, Meyer JH, Verhoeff NP. (2007). PET and SPECT imaging in psychiatric disorders. Canadian Journal of Psychiatry 2007; 52(3):146-157.
23. Wright JH. Combined cognitive-behavior therapy and pharmacotherapy. Journal of Cognitive Psychotherapy 2007; 21(1):3-6.
24. Anonymous. Folate for depression. Harvard Mental Health Letter 2007; 23(9):5.
25. Anonymous. Treatment with SSRIs linked to fractures in people over 50. Nursing Standard 2007; 21(27):14-17.
26. Inadomi JM. On-demand and intermittent therapy for gastro-esophageal reflux disease: Economic considerations. PharmacoEconomics 2002; 20(9):565-576.
27. Leontiadis GI, Virender KS, Colin WH. Proton pump inhibitor therapy for peptic ulcer bleeding: Cochrane collaboration meta-analysis of randomized controlled trials. Mayo Clinic Proceedings 2007; 82(3):286-296.
28. Durner E, Rea RS. Preventing stress-related mucosal disease in the intensive care unit. Internet Journal of Advanced Nursing Practice 2007; 8(1) Article #15236064. Retrieved April 15, 2007, on http://0-web.ebscohost.com.aspen.ald.lib.co.us/ehost.
29. Parsons ME, Ganellin CR. Histamine and its receptors. British Journal of Pharmacology 2006; 147(S1):S127-S135.
30. Parshad R, Hazrah P, Kumar S, Datta GS, Ray R, Bal S. Effect of preoperative short-course famotidine on TILs and survival in breast cancer. Indian Journal of Cancer 2005; 42(4):185-190.
31. Ashida Y, Denda M, Hirao T. Histamine H1 and H2 receptor antagonists accelerate skin barrier repair and prevent epidermal hyperplasia induced by barrier disruption in a dry environment. Journal of Investigative Dermatology 2001; 116(2):261-265.
32. Stewart J, Key AB. Histamine increases histidine uptake by basophils. Clinical and Experimental Immunology 1980; 40(2):423-426.
33. Neri PG, Stagni E, Filippello M, Camillieri G, Giovanni A, Leggio GM, Drago F. Oral Echinacea purpurea extract in low-grade, steroid-dependent, autoimmune idiopathic uveitis: A pilot study. Journal of Ocular Pharmacology and Therapeutics 2006; 22(6):431-436.
34. Goto M, Hanley KP, Marcos J, Wood PJ, Wright S, Postle AD, Cameron IT, Mason JI, Wilson DI, Hanley NA. In humans, early cortisol biosynthesis provides a mechanism to safeguard female sexual development. Journal of Clinical Investigation 2006; 116(4):953-960.
35. Kam PCA, Yarrow M. Anabolic steroid abuse: physiological and anaesthetic considerations. Anaesthesia 2005; 60:685-692.
36. Kocis P. Prasterone. American Journal of Health-System Pharmacy 2006; 63(22):2201-2210.
37. Martini LA, Wood RJ. Vitamin D status and the metabolic syndrome. Nutrition Reviews 2006; 64(11):479-486.
38. Zhou CC, Assem M, Tay JC, Watkins PB, Blumberg B, Schuetz EG, Thummel KE. Steroid and xenobiotic receptor and vitamin D receptor crosstalk mediates CYP24 expression and drug-induced osteomalacia. Journal of Clinical Investigation 2006; 116(6):1703-1712