Tales From the Swamp
by Larry Burnett, DDS
Imagine a dark, hot swamp-like world. The stench of sulfur compounds permeates the environment. Somewhere in the distance, you hear rumbling as methane bubbles churn. The stagnant fluid is rich in complex organic molecules. To survive in this hostile environment, primitive single-cell organisms somehow learn to act in concert. Different species congregate and attach to surfaces forming mushroom-like colonies. Then these single-cell creatures start demonstrating some truly strange organizational behavior.
Is this a paragraph from a Ray Bradbury sci-fi thriller? Nope. Maybe something Carl Sagan wrote speculating about complex life forms on one of Jupiter's moons? Closer ... but not quite.
It's what's going on in your patient's mouth when she sits down at a recall appointment. And once she leaves your chair, the process of recolonization starts again — even before she's reached her car.
But I'm getting ahead of myself.
In past articles, I've discussed some of the ways dental hygiene has evolved since the discovery in the late 1960s that periodontal disease is primarily a bacterial infection. When hygienists began viewing pathogens as the ultimate enemy (Not pockets. Not calculus), periodontal treatment became a lot more predictable. In fact, most cases of periodontitis are easy to treat today.
The cases that don't respond are usually complicated by one of three problems:
• An inadequate immune system
• Wretched personal hygiene
• "Hidden" bacteria that our therapy isn't reaching
For example, the culprits may have burrowed into the soft tissue, where they serve as a locus of reinfection. Or there may be a perio/endo lesion where the pocket and tooth serve as a dual source of mutual reinfection. Or (the topic of this article) the pathogens may be hiding in self-constructed bunkers.
When you read about "biofilm" in dentistry, it's usually in an article discussing how difficult it is to clean dental water lines. Flushing the water system with an antimicrobial agent can easily destroy free-floating bacteria. But once those bacteria have time to organize into a biofilm, antimicrobial agents are virtually useless.
The best known biofilm in dentistry isn't in your water lines. It's in your patient's mouth.
Bacterial plaque as a biofilm
I recently was fortunate enough to be asked by Lippincott to review preliminary copy for a book titled, "Periodontal Concepts for the Dental Hygienist" by Jill Nield-Gehrig, RDH, MA. (It's scheduled for publication in October 2002, and I highly recommend it.) Ms. Nield-Gehrig provides an excellent discussion of the biofilm called "plaque," and I've borrowed freely from her chapter on plaque development, as well as several research articles on the subject.
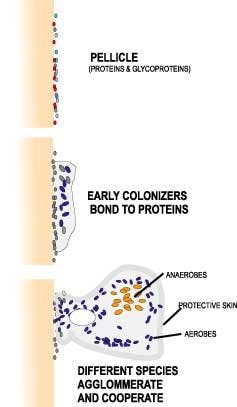
You can almost (not quite, but almost) think of plaque as a living creature in which the bacteria behave more like the specialized cells in a higher organism than as independent life forms. Let's go back to Mrs. Green again as she walks to the car after her prophy. By the time she starts the engine, those meticulously cleaned teeth are absorbing a molecular layer of proteins and glycoproteins from the saliva. This creates a surface that's ideal for bacterial attachment.
The first colonizers (primarily streptococci and actinomyces) connect to the protein pellicle and form the "roots" that will anchor the biofilm to the tooth. Fusobacteria serve largely as structural members. They promote co-aggregation of even more cells. As the biofilm develops, micro environments form inside the structure. Some areas are oxygen-deficient and are populated by anaerobic bacteria. In other areas, variations in pH occur that appeal to specific species.
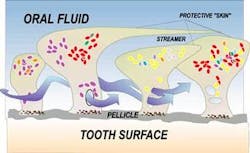
Although plaque appears to be a continuous slimy film, each micro colony really has a mushroom shape, which is connected to the tooth at a narrow base formed by those initial settlers. It branches outward as the other bacteria congregate. It's the vast number of these microscopic colonies that create the illusion of a continuous film. As the mushroom develops, weird stuff starts happening. Incompatible bacteria start living in intimate contact that would be impossible in a free liquid environment. The metabolism of the bacteria changes. They even begin communicating via chemical signals.
A slimy "skin" forms over the colony. This skin not only protects the bacteria from the hostile environment, but also seals off the good stuff like oxygen and nutrients. So tunnels form through the structure to serve as primitive alimentary canals. They allow the exterior "soup" to flow through the colony so nutrients can be absorbed and distributed ... and metabolic waste eliminated. In fact, the waste from one colony may be used by a different colony downstream.
The film factor
If periodontal disease is a localized bacterial infection (and we all know it is, right?), why aren't antimicrobial agents and antibiotics more effective in treating it? Many of the early lab studies into antimicrobials and periodontal pathogens were conducted using free bacteria in Petri dishes. When you can catch bacteria in the open, they're highly vulnerable to chemical medicaments.
But it's been estimated that 99 percent of all bacteria on earth exist as biofilms. And bacteria in a biofilm become highly resistant to antimicrobial agents. Whether it's in your unit's waterline or in a 6mm pocket, bacteria in a biofilm will withstand high pH levels, antibiotics, or antimicrobials that would be 100 percent lethal if the bacteria were exposed on their own.
In fact, studies have found that some antibiotics that kill free-floating bacteria must be increased from 50 times to as much as much as 1,500 times (1,500 times!) to be effective against the same pathogens hunkered in a biofilm.
As Nield-Gehrig observes in her book, antibiotics at these high dosages would be just as lethal to the patient as to the microbes.
And antimicrobial irrigants, like chlorhexidine or povidone iodine, don't fare much better than antibiotics. If bacteria are suspended in liquid or if a biofilm is very young (say, six hours old), chlorhexidine is fairly effective. But in a biofilm that's older (say, 48 hours or 72 hours) ... forget it! One typical study found that a five-minute exposure to 0.2 percent chlorhexidene had very little effect on the viability of a mature biofilm.
Don't misunderstand me. I'm not arguing that irrigation or antibiotics are not effective therapies for treating periodontal disease. But they must be considered as adjunctive therapies — not primary therapies. The most destructive of all perio pathogens are those loosely adherent or free-swimming anaerobes, and they're probably vulnerable to antimicrobial irrigants like iodine. Antibiotics like doxycycline may be indicated in refractory cases where the pathogens have invaded the lining of the sulcus.
But for biofilms, you need a different strategy.
Debride, debride, debride
You can't poison a biofilm. Its defenses are too good for the pharmaceutical industry.
Mechanical removal remains the only way to eliminate a biofilm. And that's one reason why debridement remains the soul of all effective periodontal therapy — both definitive treatment and maintenance therapy. In my view, the very best "hands-on" tool for removal of subgingival biofilm is the ultrasonic scaler. When I debride ultrasonically, I try like the devil to touch everything with the tip. I run it over the irregular root surfaces from different angles.
But I don't fool myself. No matter how thorough I am, I know I'm not touching all the biofilm. Many areas of the root that are accessible to bacteria are not accessible to me.
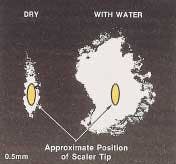
Years ago, Professor Anthony Walmsley conducted some fascinating research that showed that the power imparted to the irrigant by an ultrasonic scaler is strong enough to break dental plaque's grip on the tooth. That is, Walmsley found that ultrasonic debridement removed plaque in areas the tip didn't actually touch.
It isn't clear exactly why this works. It may be the "ultrasonic streaming" (that's a technical name for the fluid turbulence created by the high-frequency vibration). Or as Walmsley believed, it may be the energy released when the bubbles produced by ultrasonic vibration collapse (called "cavitation").
It doesn't really matter. The bottom line is this: if there's a tool that helps me remove the biofilm I overlook or can't reach with a conventional hand instrument, that's the tool I want to use.
When it was originally published, the importance of Walmsley's work wasn't fully appreciated because the treatment of perio disease lagged substantially behind the research. But as it gradually caught up during the 1990s, subgingival ultrasonic debridement became one of the major hygiene trends of the decade.
A new look at irrigation
There's also evidence that, when it's combined with ultrasonics, antimicrobial agents become more effective in treating severe periodontal cases. Again, I don't claim to know why. It may be that the ultrasonic forces the antimicrobial through the biofilm skin. Or perhaps the ultrasonic lavage breaks up the colony and gets the bacteria running so they're more easily poisoned. But that's pure speculation.
It appears that the effectiveness of ultrasonic antimicrobial irrigation depends on the severity of the disease. Mild-to-moderate pockets respond well to either water or antimicrobial in an ultrasonic lavage. It's the severe cases with 7mm pockets where iodine seems most effective.
A recently-published study by researchers in Gothen-burg, Sweden, compared the long-term outcomes of conservative treatment with and without the ultrasonic application of povidone iodine.
They selected 223 patients with advanced destructive periodontitis. To qualify for the study, at least two teeth in each quadrant had to have pockets of 6mm or more and demonstrate attachment loss exceeding 40 percent.
All of the patients were given a dog-and-pony show concerning hygiene. (Incidentally, throughout the study, a strong dose of additional education was administered whenever a patient seemed to be backsliding.) All of the patients received one phase of ultrasonic nonsurgical therapy followed by 12 years of ultrasonic supportive therapy every three to four months.
The control group and the test group received exactly the same treatment. The one small exception was that, in the test group, the irrigant used during ultrasonic debridement was 0.1 percent povidone iodine. In the control group, it was water.
Periodontal health data was collected frequently during the definitive treatment period at 0, 3, 5, and 12 months, as well as after 3, 5, and 12 years of maintenance.
Both groups showed improved gingival health, reduced pocket depth, and probing attachment. (Let's hear it for ultrasonics!) But the group that also received iodine irrigation had significantly shallower pockets and significantly greater attachment gains at every checkpoint during the initial treatment phase. That is, they got better faster. Of course, not all of the cases were successes. During years 1-3 of maintenance about 18 percent of the patients had to be referred for retreatment. However, these "losers" were not evenly distributed. Roughly 25 percent of the "scaling-only" patients had a relapse — just 13 percent of the "scaling+iodine" patients.
In the words of the authors: "The findings from the present study demonstrate that topical application of 0.1 percent povidone iodine, used as a cooling liquid in conjunction with ultrasonic subgingival root debridement, established conditions which improve the outcome of non-surgical therapy."
By the way, as in earlier studies, these researchers noted that ultrasonic administration of iodine was particularly effective in the deeper pockets.
In any war (and that includes the war against periodontal disease), it helps to understand your enemy. Most of the traditional techniques for treating bacterial infection simply do not apply to bacteria hunkered down in a biofilm. Those mushroom-shaped colonies are extremely resistant to chemicals and medicaments. That's why mechanical debridement remains the soul of effective periodontal therapy. Everything else (surgery, irrigation, systemic antibiotics, etc.) is adjunctive.
And that's why I'm such a fan of subgingival ultrasonics. By creating fluid turbulence and cavitation, that vibrating tip mechanically blasts biofilm from surfaces even beyond those I actually touch! Furthermore, there's growing evidence that when I deliver an antimicrobial in the ultrasonic lavage, I can improve the prognosis for deep pockets.
References available upon request. This article first appeared in Parkell Today.
Larry Burnett, DDS, has authored numerous articles and lectured extensively on conservative periodontal therapy throughout the United States and Canada. A frequent speaker at the ADA annual scientific session, Chicago Midwinter, and Academy of General Dentistry meetings, Dr. Burnett is featured in the video-based study program "Advanced Ultrasonics in General Practice." He can be contacted at [email protected].