Lichen planus pemphigoides: Diagnosis should be followed by referral
BY NANCY W. BURKHART, BSDH, EdD
Lichen planus pemphigoides (LPP) is a rare, blistering disease that exhibits clinical and histological features of lichen planus but subsequently develops a subepithelial separation at the basement membrane junction that is characteristic of bullous pemphigoid or mucous membrane pemphigoid. Only about 80 cases have been reported in the world's literature, but the condition is probably much more common than previously suspected.
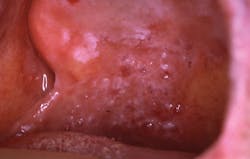
LPP has usually been reported to affect the skin, but oral mucosal lesions have been identified as well. It is usually found in adults in the fourth to fifth decade of life, although it has been reported in children and young adults too. Bullous LPP lesions may appear on the normal skin surfaces, but they may also occur on preexisting lichen planus lesions (see image for the oral manifestations).
----------------------------------------------------------
Other articles by Burkhart
- Reduction of tooth stains: Patient education is vital to preventing tooth stain
- Burkhart: Is it an oral cancer exam or screening?
- Oral Pemphigus and Pemphigoid: Why are these conditions so hard to diagnose?
----------------------------------------------------------
LPP is classified as an autoimmune disease, and it is important that health-care workers are aware of the disease and its clinical characteristics. Recognition needs to occur in the early stages of any blistering skin disease so that it may be treated in a timely manner.
LPP may be clinically or histologically diagnosed as lichen planus unless other more sophisticated diagnostic tests are performed. Typically, LPP may be more erosive, persistent, and very painful.
Bullous skin diseases such as pemphigoid, as a separate entity, have increased in Germany and are cited as occurring because of an increase in the aging population and also because of improved diagnostic procedures (Schmidt et al. 2011). These increases may add to the dilemma and confusion in reaching a conclusive diagnosis with these types of blistering skin diseases.
Diagnosis
Accurate identification requires specific laboratory tests to determine the diagnosis. The gold standard for diagnosing a blistering skin disease is immunofluorescence testing for circulation of specific autoantibodies. However, not all blistering lesions produce circulating antibodies early in the disease process. Consequently, a biopsy with microscopic examination and additional use of immunofluorescence evaluation may be necessary.
Courtesy of Drore Eisen, MD, Cincinnati, Ohio.
A study by Arbache et al. (2014) reported the results of immunofluorescence testing as a diagnostic tool for blistering skin diseases. They concluded that there was a higher sensitivity for indirect-immunofluorescence evaluation using salt-split skin technique. IIF was enhanced in the study using the salt-split skin technique that can distinguish between lichen planus or pemphigoid and facilitate LPP diagnosis.
Each type of blistering skin disease reacts in various ways with regard to deposition of the lgA/lgG components. In LPP, the location of a specific antigen (BP180 and/or BP230) on the connective tissue side of the basement membrane is characteristic for any type of pemphigoid. Laboratory tests of this type are not usually requested because the tests are expensive and consequently not cost effective on many occasions. Most clinicians who are knowledgeable about blistering skin diseases may request the above-mentioned clinical tests.
Reports have risen in the past linking LPP to treatment photochemotherapy with psoralen and ultraviolet photo chemotherapy. Ultraviolet A therapy (PUVA), cinnarizine, captopril, anti-tuberculosis therapy, ramipril, Chinese herbs (Xu et al. 2008) and simvastatin have been reported in the literature (Chan et al. 2011). It is thought that there is an inflammatory response that causes a released antigen to induce an autoimmune response against the antigen.
In the study by Chan et al., the NUVB (narrowband ultraviolet B therapy) used in the treatment of guttate psoriasis was thought to have caused antibody damage to the basal keratinocytes and progression to LPP. The authors cite that the patient was initially misdiagnosed with psoriasis.
Treatment
LPP tends to affect the lower back, buttocks, palms, scalp, and knees. The violaceous planar papules and plaques may have a silvery scale appearance as well. Histological examination will find epidermal blistering that is occurring in the specimen. With any blistering skin disease, scarring and pigmentation may occur; therefore, early treatment is essential.
The image presented depicts oral maculopapules with a mixture of both plaque-like areas and bullous vesicles. Note the reticular pattern with webbing in various areas of the tissue. The vesicles should alert the clinician to the possibility of bullous lichen planus, pemphigus vulgaris, or LPP.
As with other blistering skin diseases, topical or systemic corticosteroid therapy is often prescribed. Identifying any responsible agent is necessary and specific identification of the type of skin disease is needed. As mentioned within this column, there can be more than one skin disease presented in a patient's case.
Health-care providers are often presented with a wide and varying set of clinical features when evaluating skin lesions with or without oral manifestations. The gold standard for this differentiation is a biopsy examination often including immunofluorescence for the differentiation of the blistering mucocutaneous lesions.
Referring the patient to someone knowledgeable in the diagnosis and treatment of blistering skin diseases is needed. Clinicians treating these patients will have access to the laboratory tests that are needed to make a correct diagnosis and they are knowledgeable about the current treatment options. Usually a dental school with facilities such as stomatology centers is optimal, depending upon the location in the world. Oral pathologists and oral medicine specialists will be able to treat these patients as well.
Again, early treatment will usually result in less scarring, less anxiety, and less skin pigmentation for the patient.
As always, continue to listen to your patients and always ask good questions. RDH
References
1. Arbache ST, Nogueira TG, Delgado L, Miyamoto D, Aoki V. Immunofluorescence testing in the diagnosis of autoimmune blistering diseases: overview of 10-year experience. An Bras Dermatol. 2014 Nov-Dec; 89:6: 885-889.
2. Chan WMM, Lee JSS, Theng CST, Chua SH, Oon HWB. Narrowband UVB-induced lichen planus pemphigoide. Dermatology Reports; 2011, 3:e43. 96-98.
3. Schmidt E, Zillikens D. The diagnosis and treatment of autoimmune blistering skin diseases. Deutsches Arzteblattt International Dtsch Arztebl Int. 2011; 108:23.309-405. DOI: 10.3238/arztebl.2011.0399
4. Xu HH, Xiao T, He CD, Jin GY, Wang YK, Gao XH, Chen HD. Lichen planus pemphigoides associated with Chinese herbs. Clin Exp Dermatol. 2009 Apr; 34(3): 329-32.
5. https://www.rarediseases.org/rare-disease-information/rare-diseases/byID/44/viewAbstract
6. International Pemphigus & Pemphigoid Foundation. Sacramento, Calif. Home page: http://www.pemphigus.org. Awareness Campaign: http://www.pemphigus.org/awareness.
NANCY W. BURKHART, BSDH, EdD, is an adjunct associate professor in the department of periodontics, Baylor College of Dentistry and the Texas A & M Health Science Center, Dallas. Dr. Burkhart is founder and cohost of the International Oral Lichen Planus Support Group (http://bcdwp.web.tamhsc.edu/iolpdallas/) and coauthor of General and Oral Pathology for the Dental Hygienist. She was a 2006 Crest/ADHA award winner. She is a 2012 Mentor of Distinction through Philips Oral Healthcare and PennWell Corp. Her website for seminars on mucosal diseases, oral cancer, and oral pathology topics is www.nancywburkhart.com.