Caries prevention: Practical products and proven approaches
According to Merriam-Webster, the definition of prevention is “the act of preventing or hindering.”1 Caries is a condition that can be prevented, as it is the symptom of a disease. It is the endpoint of the infection of cariogenic bacteria and the action of that infection on the body. When thinking about caries risk management, the goal is preventing caries from developing at all.
It is true that preventing is better than curing. This is not a novel idea—in fact, Thomas Edison has been credited with saying, “The doctor of the future will give no medication, but will interest his patients in the care of the human frame, diet, and in the cause and prevention of disease.”2 A very wise statement. Yet, as dental professionals, we continue to struggle with prevention. Caries is alive and well, and in certain sectors of the population, it is rising in prevalence.3 Some may say it is even rising in severity. In order to stop caries, preventive protocols and modalities must be provided to our patients at the earliest opportunities.
Primary–primary prevention, that which is provided to the expectant parents, is both the truest form of prevention and the most effective.4 For an expecting couple, it is paramount to offer caries education, preventive strategies, and protective factors before the child arrives. This keeps the child from being exposed to cariogenic bacteria. This is critical, as early acquisition of cariogenic bacteria is a major risk factor for future caries.5–7
The opportunity to provide primary prevention can be challenging, and despite our efforts, infection by cariogenic bacteria can still occur. Given this, much of our preventive effort must rely upon controlling the highest risk factors. This includes evaluating patients’ diets, as well as counseling on oral hygiene, managing exposure to fluoride, use of calcium and phosphate, and applying sealants.
We will now look in more detail at some of the most effective caries prevention modalities—remineralization of enamel, exposure to fluoride, use of xylitol, and placement of sealants—and a group of products that can be used as part of these modalities.
Remineralization
Caries prevention starts with enamel integrity. Enamel is made up of a form of hydroxyapatite (HA) that consists of calcium, phosphate, and a hydroxyl end-member. HA is known to dissolve at a pH of 5.5, a point known as the “critical pH” of enamel.8 This means that dissolution begins at pH 5.5 and when enamel is exposed to bacteria and a fermentable carbohydrate source for those bacteria. When this happens, calcium and phosphate precipitate, resulting in a white-spot lesion. If left unchecked, the caries cycle continues and a frank lesion follows.
Enamel will demineralize with a loss of calcium and phosphate. Alternatively, it will remineralize with an abundance of calcium and phosphate. As described by John Featherstone, PhD, there is a remineralization-demineralization balance in our mouths.9 Factors that increase the possibility of demineralization increase caries risk, and factors that enhance remineralization decrease caries risk. Enamel integrity is affected accordingly.
As described by Colin Dawes, BDS, PhD, whom I had the honor of learning from as a young dental student, there can, in fact, be variation in the critical pH of enamel. Dr. Dawes notes, “The critical pH does not have a fixed value, but rather is inversely proportional to the calcium and phosphate concentrations in the solution.”10 Additionally, when teeth that have lesions are treated at an early stage (that is, prior to loss of tooth structure), they can be remineralized.10 Thus, if our goal is to maintain the mineral content of HA, calcium and phosphate must be available for uptake into the enamel.
There are a number of products on the dental market that provide calcium and phosphate to remineralize enamel. Some examples include:
• nano-hydroxyapatite,
• amorphous calcium phosphate,
• casein phosphopeptide and amorphous calcium phosphate in combination, and
• calcium sodium-phosphosilicate (Novamin).
Fluoride
The composition of HA can be changed if enamel is exposed to and uptakes fluoride, as the hydroxyl end-member is substituted with a fluoride ion. This changes HA to fluorapatite or fluorhydroxyapatite.13,14 In doing so, enamel becomes harder and more resistant to acid attack. The critical pH changes from 5.5 to 4.5, allowing for an extra “degree” of acid attack before dissolution occurs.
Fluoride exposure in the form of fluoride varnish has been accepted as highly effective, particularly in children. Compared to sound enamel, demineralized enamel uptakes a greater amount of fluoride. The mechanism of action for uptake is twofold. First, firmly bound fluoride is incorporated onto tooth surface structure. Second, loosely bound fluoride, which has the ability to act as a reservoir and enhance mineralization of adjacent tooth structure, reduces the solubility of tooth mineral and therefore inhibits demineralization.15,16
Studies show that fluoride varnish application is effective in reducing caries. Fluoride varnish is most effective when applied biannually and in conjunction with dietary management and oral hygiene counseling for at least two years. These approaches begin no later than one year of age.17,18
Xylitol
Xylitol is a five-carbon sugar alcohol derived primarily from forest and agricultural materials. Xylitol, as well as other sugar alcohols, is not readily metabolized by oral bacteria, and thus is considered a noncariogenic sugar substitute. Xylitol studies show varying results in the reduction of the incidence of caries. The famous Turku studies showed a dramatically reduced decayed, missing, filled score (DMF) for xylitol gum chewers versus sucrose gum chewers.22 Because xylitol is not fermented by bacteria, acid is not produced, and therefore pH is not decreased, demineralization is prevented, and remineralization is enhanced. Over time, xylitol can reduce the accumulation of plaque on the surface of the tooth, resulting in the bacterial makeup of the plaque changing and allowing for fewer and fewer cariogenic bacteria to survive. Use caution, however, as xylitol can cause upset stomach in some patients and is lethal for dogs.
Dental sealants
Dental sealants are an effective tool for preventing caries. They physically obstruct pits and fissures, preventing penetration and colonization of cariogenic bacteria and any fermentable carbohydrate food source.
The American Academy of Pediatric Dentistry and American Dental Association recommend the following guidelines for sealants:
• placement on surfaces based upon patient’s risk,
• careful cleaning of pits and fissures without removal of any appreciable enamel,
• diligent follow-up care,
• use of a bonding layer, and
• use of glass ionomer sealants as transitional sealants.23
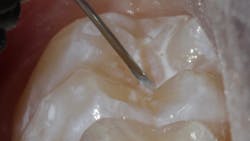
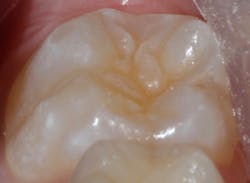
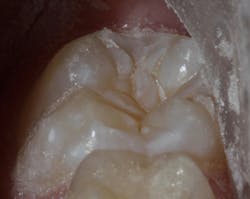
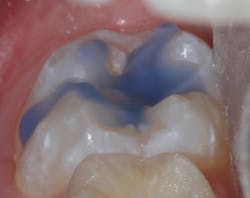
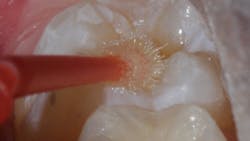
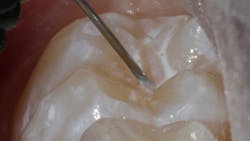
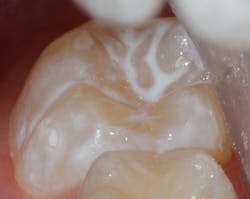
Sealant placement techniques should include complete isolation and an intermediary bonding layer (figure 1).25–29 The bonding layer and sealant curing can be one of two different methods: either placement and light-curing simultaneously in a simplified sealant technique or separately. Evidence-based studies show results favoring the individual light-curing technique in cases where conditions are dry and noncontaminated. In cases where there is saliva contamination, bond strength was the same in either simultaneous or individual light-cure placement methods.30–31
Summary
When patients have achieved a cavity-free checkup, it is gratifying for them and rewarding for us. As dental professionals, we have a toolbox available to help patients achieve and maintain health. Great products, combined with education and motivation, can give us the results our patients deserve.
References
Carla Cohn, DMD, is a general dentist who has devoted herself solely to the care of pediatric patients. Her private practice, Kids Dental, is focused on the growth and development of a cavity-free generation. She is also a part-time clinical instructor at the University of Manitoba College of Dentistry and founding member of the Women’s Dental Network in Winnipeg, Manitoba.
Disclosure: Dr. Carla Cohn is a key opinion leader for Voco GmbH. The products cited in this article are used in her course of everyday practice.
About the Author

Carla Cohn, DMD
Carla Cohn, DMD, is a general dentist who has devoted herself solely to the care of pediatric patients. Her private practice, Kids Dental, is focused on the growth and development of a cavity-free generation. She is also a part-time clinical instructor at the University of Manitoba College of Dentistry and founding member of the Women’s Dental Network in Winnipeg, Manitoba.