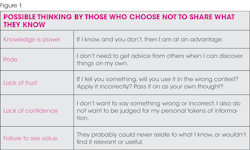
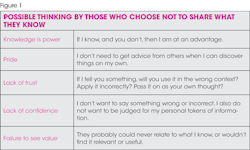
OSHA complacent or compliant?
Where does your office stand on compliance?
by GAYLENE BAKER, RDH, MBA
The dental community has been in the crosshairs after the recent events surrounding the Tulsa Okla., oral surgeon. When Dr. Scott Harrington was asked about sterilization and drug procedures in his two offices, he simply replied, "My staff takes care of that; I don't." According to OSHA, not knowing is simply not an acceptable answer. Don't be confused or complacent when complying with OSHA regulations. What exactly is OSHA, and how do dental offices comply with the guidelines?
The Occupational Safety and Health Administration (OSHA) was created in 1970 after Congress enacted the Occupational Safety and Health Act. The mission of OSHA is to ensure a safe and healthy workplace for American workers. OSHA sets and enforces standards, provides training, and promotes ongoing improvement in workplace safety and health. Its goal is to help employers protect their workers, and ultimately have fewer work-related injuries, illnesses, or deaths. Safer workplaces provide employers with reduced workers' compensation insurance costs, medical expenses, and lost work time.
Employers and employees in all 50 states and the District of Columbia, as well as other outside U.S. commonwealths, are subject to the OSHA act. However, it does not apply to some self-employed or federal agencies, or state and local governments that have their own employee safety requirements. Employers have the responsibility to provide a safe and healthy workplace that is free from serious recognized hazards. This is known as the General Duty Clause of the OSHA Act. It is the responsibility of the employer to obtain a full text of the standards, available at www.osha.gov, or by calling (800) 321-OSHA (6742). Most materials are available at no charge. With that said, it can be time consuming and cumbersome to navigate through the OSHA website. It is filled with every regulation that pertains to every business, not just dental offices. There are programs and/or services available to help dental businesses implement the appropriate regulations.
Bloodborne pathogens (29 CFR 1910.1030)
The Occupational Exposure to Bloodborne Pathogens Standard was created in 1992 and was revised in 2001 to include sharps safety. Its primary purpose was to prevent the spread of bloodborne infections. Initially the standard was aimed at hospitals, funeral homes, nursing homes, clinics, law enforcement agencies, emergency responders, and HIV/HBV research laboratories. However, the standard also covers anyone who can "reasonably expect to come in contact with blood or potentially infectious materials" as part of their jobs. Therefore, dental offices must comply with the Bloodborne Pathogens Standard. According to the Centers for Disease Control and Prevention (CDC) Guidelines for Infection Control in Dental Health Care Settings 2003, an exposure can occur in the following way:
"Exposures occur through percutaneous injury (e.g., a needlestick or cut with a sharp object), as well as through contact between potentially infectious blood, tissues, or other body fluids and mucous membranes of the eye, nose, mouth, or nonintact skin (e.g., exposed skin that is chapped, abraded, or shows signs of dermatitis)."
OSHA has not attempted to list every occupation where an exposure to bloodborne pathogens is possible. However, it does have in place a Good Samaritan act to protect employers if an employee comes into contact with bloodborne pathogens as a result of providing care as a Good Samaritan. For example, if an employee is providing first aid or CPR as a Good Samaritan and is not trained as a first responder designated to perform first aid by the employer, that employee is not covered by the standard.
Whether a dental office has two or 200 employees, the following list includes the requirements that normally apply to complying with 29 CFR 1910.1030. Annual training is required on all elements of the Bloodborne Pathogen Standard:
- A written exposure control plan
- Use of standard precautions
- Consideration, implementation, and use of safer engineered needles and sharps
- Use of engineering and work practice controls and appropriate personal protective equipment (gloves, face and eye protection, gowns)
- Hepatitis B vaccine provided to exposed employees at no cost
- Medical follow-up in the event of an "exposure incident"
- Proper containment of all regulated waste
- Needle Safety and Prevention Act of 2000
- Employee training on bloodborne pathogens and transmission prevention
- Recordkeeping
The Bloodborne Pathogen Standard is the OSHA regulation that is the most comprehensive for dental offices. Here is a brief description of the elements of the Bloodborne Pathogen Standard.
A written exposure control plan must be in place and updated annually or more often if necessary. An "exposure" is the term OSHA uses when an employee can potentially or does come into contact with a bloodborne pathogen, such as a splash in the eye from contaminated ultrasonic spray. The plan requires employers to identify, in writing, any duty or work-related activity where exposure to blood may occur. The written plan must be made available to employees and OSHA upon a visit. Standard precautions, which mean treating all body fluids as if contaminated with an infectious disease, must be followed. On the OSHA website, the term "universal precautions" is used. OSHA considers universal and standard precautions to be interchangeable.
Proper engineering and work practice controls to avoid potential exposure to a bloodborne pathogen need to be applied. Work practice controls reduce the chance of exposure by changing how a task is performed. Examples of work practice controls are hand washing techniques, procedures for handling needles, and prohibiting eating, drinking, applying lip balm, etc. in work areas where bloodborne pathogens may be present.
Engineering controls act on the source of the hazard. They reduce exposure by removing or isolating the hazard. Examples of engineering controls are puncture-resistant sharps containers, self-recapping needles, and biohazard waste containment bags. Bloodborne Pathogen Standard (29 CFR 1910.1030) also takes into account procedures to minimize needlesticks, minimize splashing and spraying of blood, ensure appropriate packaging of specimens, and regulate wastes and decontamination of equipment. Consideration, implementation, and use of safer engineered needles and sharps must include everyone in the office and be discussed annually.
Personal protective equipment (PPE) must be worn and supplied by the employer at no charge to the employee. In the dental operatory, appropriate PPE is considered to be a facemask, eye protection with side shields, medical exam gloves, and a lab gown. OSHA refers to the CDC guidelines for what constitutes the appropriate level of protection.
The hepatitis B vaccination must be made available to all employees who have occupational exposure to blood. The vaccination must be offered within 10 working days from the start of employment. The employer must pay for the vaccination, allow the vaccination to be administered during regular business hours, and this must be under the supervision of a licensed physician/licensed health-care professional. Employees can choose not to have the vaccination but must sign a declination form. They also have the right to rescind the declination form at any time and receive the vaccination at no charge.
In the event of an exposure to a bloodborne pathogen or other potentially infectious material (OPIM), a postexposure evaluation and medical follow-up must be completed. It specifies that the procedures to be made available to any or all employees who have had an exposure incident. An accredited laboratory must conduct lab tests at no cost to the employee. A follow-up must include a confidential medical evaluation documenting the circumstances of the exposure, identifying and testing the source if known, testing the exposed employee's blood if he/she consents, postexposure prophylaxis, counseling, and evaluation of reported illnesses.
Labels or color-coded stickers must be placed on regulated waste or items contaminated with bloodborne pathogens. Examples are sharps containers, biohazard containers, and contaminated laundry. Orange or red-orange labels containing the biohazard symbol must be affixed to the containers for these items. Red bags or containers may be used instead of labeling. When a facility uses standard precautions, the laundry does not need any special labeling. Refrigerators and freezers or other containers used to store items that may be contaminated must display the biohazard sign.
Employees must be trained within 90 days of effective hire date, initially upon assignment, and then annually. Employees who have received appropriate training during the past year need only receive additional training in items not previously covered. Employees must be trained on bloodborne diseases and their transmission; the exposure control plan; engineering and work practice controls; personal protective equipment; hepatitis B vaccine; response to emergencies involving blood; how to handle exposure incidents; the postexposure evaluation and follow-up program; needle safety and prevention, including safer handling of sharps and evaluation of safer sharps by all employees; and signs/labels/color-coding. There must be an opportunity for questions and answers, and the trainer must be knowledgeable in the subject matter.
Recordkeeping standards are for all medical records and should be kept for each employee with occupational exposure risk for the duration of employment plus 30 years. Training records must be maintained for three years and must include dates and the details of training.
Hazard Communication Standard (29 CFR 1910.1200)
The Hazard Communication Standard (HCS) is sometimes called the employees' right-to-know standard. It requires that the employees be informed and made aware of hazards and have access to safety data sheets (SDS, formerly known as MSDS) of all types of chemicals used in the facility. OSHA states an estimated 650,000 hazardous chemicals currently exist and new ones are added every year. HCS educates employees and employers about the hazards, precautions to be taken, and side effects of chemical use and exposure.
Dental offices must have a written HCS plan that lists the chemicals stored in the office, who handles the different portions of the plan, and where employees can access the plan. The written plan must also describe how the dental office will meet OSHA's requirements regarding labels, warnings, SDS, employee training, and information. Presently there is no endorsed method of labeling hazards, just that they must state what the hazard is.
In 2003, the United Nations (UN) adopted the Globally Harmonized System of Classification and Labeling of Chemicals (GHS). The GHS includes criteria for the classification of health, physical, and environmental hazards, specifying what information should be included on labels of hazardous chemicals as well as SDS. The United States was an active participant in the development of the GHS, and is a member of the UN bodies established to maintain and coordinate implementation of the system.
The GHS symbols have been assimilated into pictograms for use on the GHS label. Pictograms include the harmonized hazard symbols plus other graphic essentials, such as borders and background patterns or colors, which are intended to convey specific information. OSHA published a proposed rulemaking on September 30, 2009, to align OSHA's Hazard Communication standard (HCS) with the GHS.
The new GHS will become an OSHA standard in 2016. The standard that gave workers the right to know, now gives them the right to understand. It has taken 10 years for uniformity in chemical safety to take place; we should embrace this safety for us, the dental professional. Have you ever thought of what chemicals you work with? Be ready for this change; it's for your personal benefit.
PPE general requirements (1910.132)
OSHA refers to standards of protection that have already been written and put in place by the CDC when applicipable. Personal protective equipment (PPE) are items used to protect the dental health-care provider (DHCP) from coming into contact with a bloodborne pathogen or OPIM. According to OSHA, protective equipment, including personal protective equipment for eyes, face, head, and extremities, protective clothing, respiratory devices, and protective shields and barriers, shall be provided, used, and maintained in a sanitary and reliable condition. Training on proper use of PPE and regular evaluation needs to take place. PPE should be supplied and used properly to protect against the designated hazard.
Hazards come in many forms. They can be environmental, chemical, radiological, or mechanical irritants that when encountered in a certain way makes them capable of causing injury or impairment in the function of any part of the body through absorption, inhalation, or physical contact. The question of laundering scrubs often arises. If scrubs do not function as PPE but as a uniform, then OSHA does not have oversight on laundering. If scrubs function as PPE, then the employer is in violation of the BBP standard by having employees clean, launder, and maintain scrubs on their own (1910.1030(d)(3)(iv)). Lab jackets are PPE, and they are required to be laundered by the employer. If staff members wear only their scrubs as PPE and no lab jacket or disposable jacket, they are exposing their arms to aerosols from the handpiece, ultrasonic scaler, or air/water syringes. Now imagine someone going to lunch in contaminated scrubs. They are exposing everyone at lunch, either in an employee lounge area or restaurant, to potentially pathogenic microorganisms. Each dental facility should have a written policy on proper training and adequate PPE selection for employees.
Hand protection (1910.138)
Hands are porous and can act like a sponge. Chemicals that are not meant for the body to absorb are used on a daily basis. Wearing suitable hand protection for a task is very important. The gloves used to treat patients are very different than the gloves to be worn to process instruments. Wearing clinic gloves to process instruments is very dangerous. Thicker, chemical-resistant gloves are recommended. Instrument processing gloves are referred to as utility gloves and should be puncture resistant. They may be clumsy at first, but are the safe choice. Employers must select the appropriate hand protection and require employees to use it. Hands need to be protected from exposure to hazards such as those from skin absorption of harmful substances, severe cuts or lacerations, severe abrasions, punctures, chemical burns, thermal burns, and harmful temperature extremes. Employers should base their selection of hand protection on the performance features of the protection relative to the task(s) to be performed, conditions present, duration of use, and and the hazards and potential hazards identified.
OSHA poster
Every workplace must display the OSHA poster or the state plan equivalent. The poster explains workers' rights to a safe workplace and how to file a complaint. The poster must be placed where employees can easily see it. The OSHA poster (English 3165-2007) can be ordered or downloaded at www.osha.gov/Publications/poster.html.
Don't chance losing the professional license you worked so hard to obtain, or the business you've worked so hard to develop. If you don't think you could lose your license due to an OSHA violation, read what happened to Stephen Chadwick at www.leagle.com (Stephen Chadwick v. Board of Registration in Dentistry SJC-10831). If you need help making your dental office OSHA compliant, there are many resources to assist in the process.
Visit the OSHA website or call an OSHA office to ask questions, go to the ADA for direction, and talk with your sales rep about programs specifically designed to make compliance easy. There are also several qualified independent OSHA trainers, and many trainers are dental office specific. They will use their expertise to train dental professionals in regard to safety and awareness in their offices. A recent publication, Model Plans & Programs for the OSHA Bloodborne Pathogens and Hazard Communications Standards (Publication 3186-06R), contains a prototype of these two important documents that can be specifically tailored to each office.
Why take risks when you don't need to? OSHA is not a land far, far away, nor is it something to be afraid of. RDH
References:
CDC MMWR December 19, 2003/Vol.52/No. RR-17
http://www.cdc.gov/nceh/radiation/default.htm
GAYLENE BAKER, RDH, MBA, graduated from the University of Iowa with a bachelor's degree in dental hygiene and from Aurora University with her MBA. Over the course of her career, she has been employed in a variety of private practice settings. Currently, Gaylene is employed by Crosstex International as a Midwest Region Sales Manager. Gaylene is a member of ADHA, AADH, AAOSH, and the ADAA. She is available to speak at dental schools and dental hygiene schools, study clubs, local component meetings, and dental offices in her territory. She can be reached at [email protected].
The standards for dental offices include but are not limited to:
- Bloodborne pathogens (29 CFR 1910.1030)
- Hazard communication (29 CFR 1910.1200)
- Ionizing radiation (29 CFR 1910.1096)
- Exit routes (29 CFR Subpart E 1910.35, 1910.36, 1910.37, and 1910.38 and 1910.39)
- Electrical (Subpart S-Electrical 29 CFR 1010.301 to 29 CFR 1910.399)
- OSHA poster display (OSHA Publication 3165)
- Reporting occupational injuries and illnesses (29 CFR 1904)
The following standards, in order, were the most frequently cited offenses by OSHA from October 2010 through September 2011, in Offices and Clinics of Dentists:
- Bloodborne pathogens
- Hazard communication
- General requirements (electrical)
- General requirements (personal protective equipment)
- Eye and face protection
- Medical services and first aid
- Wiring methods, components, and equipment for general use
- General requirements (walking-working surfaces)
- Portable fire extinguishers
Source: From the OSHA website http://www.osha.gov/dsg/hazcom/effectivedates.html
Past RDH Issues