The corner in the round room: What is the objective in countering biofilm and periodontal disease?
Dental hygienists, as licensed health-care professionals, not only provide clinical care, but must also evaluate data and help the dentist make knowledge-based decisions regarding the effectiveness of treatment for patients. In my attempt to find effective solutions for patients’ periodontal concerns, I’ve always felt an obligation to investigate any promising new products and protocols.
Why? Because what we were doing in the practice simply wasn’t working predictably, yet we still kept doing the same thing. As Albert Einstein said, “Insanity is doing the same thing over and over again and expecting different results.”1
As a profession, we have to stop following the scale, polish, drill, fill, and bill routine. Shouldn’t we understand and treat the cause, not just the symptoms of periodontal disease? This is not a new concept! In fact, the first Chinese medical text from 2600 BC states, “Superior doctors prevent disease. Mediocre doctors treat disease before it is evident. Inferior doctors treat full-blown disease.”2
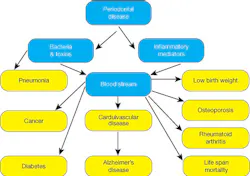
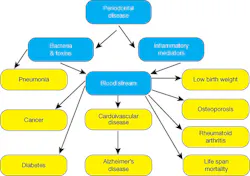
Where does your practice fall in this description, and where do your patients want you to be? The new paradigm is the practice of oral medicine, with the focus on preventive interception before disease is evident in order to avoid the consequences of poor oral health. This is a huge responsibility. When we consider that periodontal disease is accepted as causative for Alzheimer’s disease, 50% of all heart attacks/strokes, and several forms of cancer, then clearly, we need to stop the bleeding.3
Are you tired of looking for the corner in the round room? Are you ready for real change? Remember, our patients expect us to provide the most competent therapies available, and it is our clinical and fiduciary duty to do exactly that. Can you provide answers and solutions to the following questions?
- Why do 75% of patients have periodontal disease (defined as bleeding when tissues are touched) in spite of our best efforts?
- Why don’t patients know how they catch and spread periodontal disease? It is hard to prevent a disease if you don’t know how it originates.
- Does cleaning teeth (removing calculus and stain) twice a year really prevent periodontal disease, especially if a patient still has bleeding on tissue manipulation?
- Patients won’t floss in spite of our best efforts. Is flossing the best home-care technique to clean interproximal spaces to prevent periodontal disease, or are there better methods? What are they?
- Is brushing your teeth the best thing to do if you have gum disease? Are you cleaning in the wrong places?
- Why can’t we predict which areas are infected and will bleed prior to scaling? Think about this. Does a surgeon start surgery without knowing if there is a clotting disorder or if infection is present?
- Why don’t we treat periodontal disease as a biofilm infection, using medical disinfection principles and protocols that are the standard of care in all other medical professions?
- How do we introduce the oral-systemic link concept to patients so that they understand its importance?
First and foremost, we need to use protocols that are scientifically proven to work in order to educate our patients about the association of periodontal disease with systemic diseases. We need to demonstrate the presence of disease visually before we start a therapy appointment. Quite simply, we have to stop doing “bloody prophies” and accepting them as the norm.
Know the enemy: biofilm
Our new goal for every patient must be excellent biofilm control and zero bleeding with both home care and periodontal therapy. With the evident oral-systemic connection, we need to routinely test for the presence of periodontal pathogens, such as T. denticola and P. gingivalis, and consider the use of new therapies, such as antibiotic rinses, to kill and control these pathogens.
As a profession, we also need to address the fact that our traditional methods of treating the oral infections that cause periodontal disease and halitosis have not been working consistently, and that patients do not always accept the responsibility of keeping their mouths healthy. Remember that patients have to look after their mouth 365 days a year, 24 hours a day, but we see them for only three to four hours a year to help coach, motivate, and provide periodontal care. Scaling alone doesn’t work because it doesn’t control bacteria.
Patients have to know that you care about their health; therefore, at every visit try to show and teach them something new. When you use this approach, patients will perceive it as encouraging and focused on promoting oral and overall health, and not as selling or pushing periodontal therapy.
Figure 1
Source: The oral-systemic science: emerging evidence of a strong relationship. Oral Health. December 2010.
According to Dr. Wei et al., “Gingivitis and mild periodontitis are usually reversible conditions and should be detected early and treatment rendered promptly. Hence, appropriate clinical indices must be available to record these conditions accurately and to assess treatment success or failure.”4
If you told a patient they were going to lose a finger or toe, they would probably be very upset. So why do dental professionals and their patients find the loss of teeth and their subsequent replacement a perfectly normal event? We have to acknowledge that gingivitis is a condition that can be life threatening and dramatically impact health and life expectancy.5 We should be looking at why our patients are losing their teeth and change our protocols for earlier intervention so this does not happen.
The American Academy of Periodontology’s “Parameters on Refractory Periodontitis” outlines the following steps for diagnosis and treatment: “Once the diagnosis of refractory periodontitis has been made, the following steps may be taken:
- Collection of subgingival microbial samples from selected sites for analyses, possibly including antibiotic-sensitivity testing.
- Selection and administration of an appropriate antibiotic regimen.
- In conjunction with the administration of an antimicrobial regimen, conventional periodontal therapies may be used.
- Reevaluation with microbiological testing as indicated.”6
Periodontal disease exists if gums bleed, periodontal pockets are present, or possibly even if a patient simply complains of bad breath. The same bacteria that cause periodontal disease cause halitosis and have the same harmful effects on the organs throughout the body. Prior to treatment, patients should be informed about systemic risks. However, before we can educate patients about these risks, we need to educate ourselves. Our profession underestimates the seriousness of periodontal disease and the concept of biofilm load versus severity of the disease and its systemic ramifications.
Spirochetes is the link
By the time the disease is obvious, the risk factors from the oral-systemic link have been activated and are already in place. Are you aware that 93.7% of all Alzheimer’s patients’ brains harbor at least one of five species of oral spirochetes?7 Healthy brains do not have spirochetes. Syphilis is caused by a spirochete. Judith Miklossy calls Alzheimer’s a “neurospirochetosis.” All syphilitic brains contain spirochetes and their structural changes are identical to Alzheimer’s. Also, 100% of patients with Lyme disease have spirochetes in their brains.7
If you were told you had syphilis or Lyme disease, you would demand immediate treatment involving appropriate antibiotics. Periodontal disease is also a spirochete disease where people lose body parts such as teeth and alveolar bone. Why don’t we react with the same concern with periodontal disease at the earliest signs of bleeding?
Suggested location for p01
Did you know that 50% of all heart disease is caused by oral bacteria combined with the body’s reaction to them - inflammation? Are you aware that 37% of all blood clots from heart attacks and strokes contain live periodontal pathogens (Pg), and that 100% of these clots contain DNA residues of these periodontal pathogens?3 There is a huge impact on our patients’ overall health and the associated medical cost of treatment because of periodontal disease. Imagine the positive impact that effective periodontal care could have on our patients’ overall health.
All of our patients want to be healthy, but they don’t understand how to predictably achieve health or how their oral health is so intimately related to their overall health. This is an opportunity to raise your standard of care. It is important to tell your patients that periodontal disease is spread to their partners, children, and the ones they love via saliva transfer, among other ways, through kissing.
Periodontal disease is also associated with diabetes,8 preterm birth,9 several cancers,10 and many other conditions.11 According to Dr. Manuel Quinonez, “What matters is the burden poor oral health and exposure to pathogenic bacteria places on the body in determining morbidity and mortality outcomes in these systemic diseases and conditions.”12
Where there’s bleeding, there’s a wound.
Many dental practices use systems that record pocket depths and bleeding, but what do they do with this data? In order to maximize patients’ understanding of this process, we need to discuss healthy versus unhealthy numerical scores and what they mean in the context of oral and systemic health. Measurements should be done on a yearly basis for adults. During the examination protocol, hygienists should verbally communicate to patients the location of bleeding on probing (BOP) and count the number of bleeding points to give them a formal score.
Without a score, you do not have an objective test. It is important to think of bleeding as a wound that will never heal until the cause of the bleeding is stopped. Ideally, BOP should be less than 10. Why not zero? It is nearly impossible to clean 360 degrees 3 mm to 4 mm around 28 teeth perfectly. A 30 BOP wound is equivalent to a skin wound about the size of a silver dollar. If this wound was on the skin, it would get immediate emergency-room medical attention with referral as needed, especially if it did not heal. What’s more, an attending physician would not scrape it with steel wool and tell the patient to return in six months!
But isn’t this what we as dental clinicians do for periodontal disease? A wound this size is one of the major risk factors with periodontal disease and systemic concerns as this creates bacteremias that spread throughout the circulatory system on an ongoing basis. The papillary bleeding score (PBS) is done by checking for bleeding with the use of a Soft-Pick before any scaling. It serves as an emotional and diagnostic tool to identify areas of infection (bleeding) and inflammation (soreness), and to vividly demonstrate infection in the mouth before scaling. It is by far the most important test we can do to demonstrate health or periodontal disease at each hygiene appointment, and it makes it easy for patients to understand the severity of the infection.
This in turn motivates patients to accept the need for daily interproximal cleaning, more frequent hygiene appointments, microbiology assessment, and antibiotic rinses followed by a reevaluation appointment.
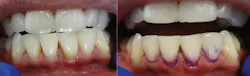
Figure 2 is an example of what appears to be a healthy mouth, and the result when the papillary bleeding test is administered.
When patients see the bleeding they routinely ask, “How do I stop the bleeding?” This question opens the door for you to end “bloody prophies.” The moderate pressure that cleans and disturbs the biofilm and granulation tissue is responsible for the bleeding. Be sure to explain to patients that healthy tissue does not bleed or hurt when touched with tools such as the Soft-Pick. Tissues shrink and go from healthy-looking beer belly gums to six-pack abs in three days. Therefore, the PBS is the “truth serum” for infection and shows whether your patients are cleaning between their teeth. This is the first and most important change to make in their daily routine.
Full disclosure on disclosing
We cannot see biofilm. However, simple low-tech disclosing clearly shows both the patient and clinician the areas where thriving biofilm colonies remain undisturbed. With disclosing, patients can easily see the trouble areas, and will become motivated to remove the biofilm and accept instruction to improve their technique.
Disclosing visually demonstrates to your patients that improvement is needed in their home-care technique. What’s more, removing the biofilm before scaling and root planing (SRP) is necessary, and disclosing should be done on all patients. Despite the value of disclosing for controlling oral biofilm, many clinicians do not like to disclose because they think it’s messy or their patients don’t want it. Sometimes we need to be assertive in doing what a patient needs, not what a patient wants. Applying the two-tone disclosing solution with a microbrush is simple and overcomes the “it’s messy” objection. I believe it’s one of the more powerful tools we can use to help patients control their disease.
As shown in Figure 3, the patient needed gingival grafting. We didn’t realize we needed to review biofilm control until he was disclosed. Good thing we did it!
You can’t effectively treat if you don’t accurately test
You should recommend biofilm testing if a patient has a significant medical history or visible signs of periodontal disease. This is done before scaling so the subgingival biofilm is undisturbed and an accurate sample can be taken. There are salivary tests from companies such as OralDNA Labs and Hain Diagnostics that provide a generalized assessment of pathogens present in the oral cavity, and the degree of infection known as bacterial load. Saliva samples are an indirect measure of subgingival biofilm and saliva sampling variations. Paper point sampling is more accurate for subgingival biofilm but it does not sample the whole mouth biofilm.
OraVital Inc. offers two different testing systems for comprehensive oral biofilm assessment. For information regarding the specific pathogens causing the infection, we found OraVital’s BiofilmDNA analysis to be the most comprehensive. Sampling for this test is easy and consists of saliva as well as supragingival, subgingival, and tongue biofilm testing using paper points and a swab. The lab report provides details on the presence of six species of periodontal bacteria, S. mutans for caries susceptibility, and Candida albicans.
Another useful test from OraVital is BiofilmGS, a gram stain analysis test that provides information on classes of bacteria, yeast, amoebae, and neutrophils. This is for site specific testing of six sites - throat area, tongue, and quadrant sampling. This test takes less than two minutes.
Microbiological reports when combined with clinical signs present in the patient’s oral cavity help to determine the type of periodontal treatment that is required, and offers suggested targeted antibiotic/antifungal rinses versus oral pills. Rinses offer a nonsystemic and more concentrated antibiotic in the saliva compared to pills. Biofilm analysis prior to treatment of periodontal disease and halitosis ensures that both clinician and patient understand the extent of infection and the risks associated with this infection.
This type of third-party verification ensures very complete patient records. The use of an objective biofilm test to determine the presence of oral pathogens is an important adjunct in diagnosis, treatment planning, and last but not least, patient acceptance.
Treatment modalities
There are numerous traditional treatment modalities for periodontal disease that include laser care, SRP, locally-delivered antibiotics such as Arestin and Atridox, as well as the use of oral-systemic antibiotics such as amoxicillin. Another highly effective alternative is the use of OraVital’s personalized and prescribed antibiotic/antifungal and antimicrobial rinses for at-home treatment following the completion of scaling and root planing or other periodontal treatments. Since these rinses are colloidal suspension liquids, the antibiotic particles enter the gingival crevice as deep as 7 mm to decrease pathogenic biofilm.vii
In our practice we also use 0.2% chlorhexidine after the antibiotic/antifungal rinse regimen to help decrease some of the Streptococci. This one-two punch has been very successful in helping our patients control both periodontal disease and halitosis. The In a published article in July 2013, the Academy of General Dentistry commented on the results obtained in a study of 649 patients. The article pointed out a reduction of BOP by 87% and pocket depths 1 mm to 2 mm when using the OraVital System for two weeks with no scaling.13 I recommend scaling, but it was not done for these patients as part of their treatment.
Evaluation and maintenance
Patients should return for a reevaluation appointment four weeks after rinses are started in order to check on the success of the treatment. By repeating the same objective tests (number of BOP, PBS, disclosing, and periodontal charting) and comparing pre- and post-treatment results, areas that have been successfully treated can be identified. For those areas that do not respond, further treatment can be prescribed. This is also a good opportunity to use disclosing solution again to check for inadequate oral self-care and to review proper technique. Maintaining good oral health through mechanical (daily home care and regular hygiene appointments) and chemical (rinsing) disruption of biofilm is necessary. Without continual biofilm disruption, oral infection will return.
As we all know, many of our patients do not floss. According to the Journal of Clinical Periodontology, “Data support the belief that professionally administered plaque control significantly improves gingival inflammation and lowers plaque scores, with some evidence that reinforcement of oral hygiene provides further benefit. Flossing cannot be recommended other than for sites of gingival and periodontal health, where interdental brushes (IDBs) will not pass through the interproximal area without trauma. Otherwise, IDBs are the device of choice for interproximal plaque removal.”14 Think about it - you can’t floss the inside of a bowl, the shape of interproximal tissues. It will not clean gingival biofilm!
Wow, flossing is not recommended with active periodontal disease! That’s why we recommend supplementing flossing with Soft-Picks and Stim-U-Dents. Your patients will love using IDB and will use them on a daily, which will have a positive impact on their home-care compliance, and ultimately, their oral health and overall health.
Remember that it is important to detect and eliminate pathogens and rebalance the biofilm for the entire oral cavity in order to maintain oral tissue health. There has to be no biofilm on disclosing, fewer than 10 BOP pockets, and less than 4 mm and no bleeding when the tissues are touched. Once treatment is completed, the patient needs to use daily mechanical and antimicrobial rinses for continued biofilm control.
Patients can feel the difference within days and see the reduced bleeding. Continuously reevaluate the results of your therapies, adopt new techniques, and enjoy the feeling of making a difference in your patients’ lives. Stop looking for the corner in the round room! Start now to offer the best protocols you can to heal your patients and stop the bleeding. RDH
Kerry Lepicek, RDH, has over a decade of experience using an interceptive and preventive approach to treating the oral infections that cause periodontal disease and breath odor. She currently practices in Toronto, and is also the director of training and education at OraVital Inc. Kerry has been published in dental industry publications such as Oral Health Magazine and has lectured at dental meetings in Canada, the United States, and Bermuda. She can be contacted at [email protected].
References
1. Albert Einstein - German Theoretical-Physicist (1879-1955)
2. Neijing H.(2600 BCE) First Chinese Medical Text, Superior Doctors: A Review of JAMA. Access to Knowledge, 4 Volume 1 (2012).
3. Pessi T, Karhunen V, Pasi P, et al. Bacterial Signatures in Thrombus Aspirates of Patients with Myocardial Infarction. Circulation. 2013;127:1219-1228.
4. Wei S, Lang KP. Periodontal epidemiological indices for children and adolescents: I. gingival and periodontal health assessments. Med Dent Pediatric Dentistry/Copyright e 1981 by The American Academy of Pedodontics/Vol. 3 No. 4 Pages 354-360
5. Soder B. Karolinska Institutet, Stockholm Sweden Opportunities for advancing dental hygiene research; Periodontitis and premature death: a longitudinal, prospective clinical trial.(Clinical report) Can J Dent Hyg, 2009, 43, N5. 217 -223.
6. Position Paper Systemic Antibiotics in Periodontics J. Periodontal 2004 75:1553-1563.
7. Miklossy J. Alzheimer’s Disease- a neurospirochetosis. Analysis of the evidence following Kosh’s and Hill’s criteria. Journal of Neuroinflammation 2011, 8:90 http://www.jneuroinflammation.com/content/8/1/90
8. Genco RJ. Opportunities for advancing dental hygiene research; Periodontal disease and association with diabetes mellitus and diabetes: clinical implications. Can J Dent Hyg. 43(5):217-223, 2009.
9. López NJ, Smith PC, Gutierrez J. Higher Risk of Preterm Birth and Low Birth Weight in Women with Periodontal Disease J Dent Res 81(1):58-63, 2002
10. Michaud DS, et al. Periodontal disease, tooth loss, and cancer risk in male health professionals: A prospective cohort study” Lancet Oncology 2008; 9:550-58.
11. Toop J, Sorci R. The Oral-Systemic Link: Monsters in Your Mouth. Inside Dentistry October 2015, Volume 11, Issue 10. https://www.dentalaegis.com/id/2015/10/the-oral-systemic-link-monsters-in-your-mouth
12. Quinonez C. The truth is out there. Ontario Dentist 4 2015: 14-15.
13. Southward K, Bosy A. Treatment of oral malodor and periodontal disease using an antibiotic rinse. General Dentistry 2013; July:41-46
14. J Clin Periodontal 2015; 42(Suppl.16): S71-S76 dio: 10.1111/jcpe 12366 Primary prevention of periodontitis: managing Gingivitis Chapple
About the Author

Kerry Lepicek, RDH
With more than 20 years of clinical experience, Kerry Lepicek, RDH, is an international lecturer on oral biofilm, halitosis, oral-systemic connection, and the dental hygiene process of care. She is the editor for The Hygiene Corner with Women in Dentistry, serves on the advisory board for the American Academy for Oral Systemic Health, and is a cast member on The RDH View. Kerry recently received the Award of Distinction from the Ontario Dental Hygienists Association. Reach her at [email protected].