What's in the water? Concerns over dental unit water lines challenge compliance efforts for patient safety
By Ellen R. Guritzky, RDH, BS
Have you ever stopped to think about the purity of a glass of water before you took the first sip? In most cases, you don't need to stop and consider if your tap water, known as potable water, is drinkable since it is presumed that governmental agencies routinely oversee these concerns prior to the water being consumed. The agencies and associations responsible for ensuring drinkable water include the Environmental Protection Agency (EPA), American Public Health Association (APHA), and American Water Works Association (AWWA). Each of these entities has strict standards for ensuring that water is safe for consumption. In the event there is a problem with water quality, notification is sent out by one of these groups and necessary steps are taken to correct the issue.1,2,3,4,5
Similar to the scenario related above is the situation encountered by patients visiting a dental office. Patients trust that the water used throughout their visit is treated with the highest level of care. In addition, patients are encouraged to ask questions in order to ensure infection-control protocols are being followed and their health safeguarded. Things patients should look out for include, but are not limited to, watching personal protective equipment being donned and doffed, instruments being unwrapped from cassettes/bags, and the application of barriers on equipment. Furthermore, they should inquire about those infection control procedures that cannot be witnessed such as weekly biological monitoring tests to assure sterility of the instruments and minimizing risk of cross contamination. Patients do not routinely think about the water that flows from a high-speed handpiece, three-way syringe (air/water syringe), or ultrasonic scaler, because it is expected that practitioners are following guidelines to keep water free of contamination. Just like the tap water they drink, they assume the water is pure. But is it?6,7,8
What's found in water lines
The Centers for Disease Control and Prevention (CDC) found that there are more than 200 species of microorganisms cultured from dental unit water lines (DUWL). Random testing of these water lines has documented colony counts that are 400 times higher than the local water supply. A wide range of microorganisms has been isolated from DUWL. These include fungi, free-living amoebae, protozoa, nematodes, and human pathogens such as Pseudomonas aeruginosa, Legionella pneumophila, Mycobacteria species, and Staphylococcus species. Colony-forming units per milliliter (CFU/mL) of heterotrophic bacteria are cultured to assure low levels. CFU is "the minimum number of separable cells on the surface of or in semisolid agar medium which gives rise to a visible colony of progeny is on the order of tens of millions. CFUs may consist of pairs, chains, and clusters as well as single cells and are often expressed as colony-forming units per millimeter."9,10,11,12
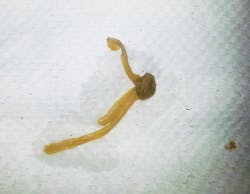
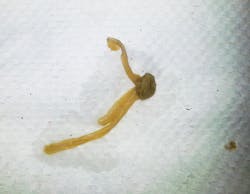
Actual photos of biofilm in dental unit waterlines.
CFU counts identify the number of viable bacterial cells in a sample per milliliter, or degree of contamination, in water, vegetables, soil, or fruits, or the magnitude of the infection in humans and animals. The CFU/mL count should not exceed 500 CFU/mL to ensure minimal heterotrophic bacteria in the water. Heterotrophs-organisms that derive nutrition from complex organic substances such as yeast, molds, and bacteria-are found in every type of water and should be kept to a minimum. Heterotrophic bacteria found in water typically pose no health risk to humans; however, a high heterotrophic plate count (HPC) is an indicator of ideal conditions of bacteria growth. A high HPC count can also be a breeding ground for more dangerous bacteria, such as Legionella or E. coli; it can cause foul-tasting water and lead to corrosion or slime growth in pipes or DUWL.13,14,15
In 1995's "Statement on Dental Unit Waterlines," the ADA recommended flushing of DUWL. However, while that procedure is still recommended, it should be noted that flushing does not completely remove biofilm from the lines. Studies have revealed that even after flushing for four minutes, the level of CFUs remaining still exceeded ADA recommendations for water quality, and biofilms continued to exist. Flushing the water lines for 20 to 30 seconds in between patients is recommended so that the number of bacteria in the water line can be decreased. But treatment is still needed to reduce CFU counts. Dental unit water that remains untreated or unfiltered is not likely to meet drinking water standards. The ADA position statement recommended less than or equal to 200 colony-forming units/mL, while in 1993, the CDC recommended less than 500 CFU/mL from a DUWL. By using water that has <500 CFU/mL of bacteria (e.g., tap, distilled, or sterile water) in a self-contained water system, bacterial contamination from the DUWL is not eliminated. Moreover, any dental devices that are connected to a DUWL, such as handpieces, ultrasonic scalers, or air/water syringes, should be purged/run for a minimum of 20 to 30 seconds after each patient use.16,17There is documented research showing that microbial counts can reach as high as 200,000 CFU/mL within five days after installation of new dental unit water line. Counts this high occur because factors such as the system design, flow rates, and materials may promote bacterial growth and development of biofilm. Dr. Shannon Mills describes it as "the perfect convergence of biology (surface colonization/attachment and proliferation of biofilms), physics (laminar flow), and geometry (surface to volume ratio)."18,19
A dental unit water line is manufactured from a polyurethane or polyvinyl chloride (PVC) tube that is about 1/16th inch (or approximately 2 millimeters) in diameter. The flow of water significantly decreases as it reaches the DUWL. Water travels in tubes, about 10 inches wide, from the city line to the 1/2-inch plumbing in the dental office, and finally to the 1/16th-inch DUWL.20,21
Publicized connections
There is a growing connection of DUWL and patient illness/fatality. In February 2011, an 82-year-old woman was admitted to the intensive-care unit with fever and pneumonia. After undergoing various tests, her diagnosis revealed that the patient had Legionnaires' disease. Immediate treatment was delivered; however, the patient developed severe, sudden, and irreversible septic shock. She succumbed to this condition and died two days later.
Upon subsequent investigation, it was found that the woman had no obvious exposure to the Legionella infection, and that she left the house only for two dental appointments. After water samples were taken from DUWL at her dental office, Legionella pneumophila was discovered. It was then determined that her infection was acquired from the DUWL during her dental treatment. Since then, reports of DUWL with microbial biofilm have been prevalent.22,23
More recently, in September 2015, it was reported by a television station in Atlanta that a number of children developed infections that were traced back to a pediatric dental clinic in Jonesboro, Ga. Children's Health Care of Atlanta reported more than a dozen individuals who were found to have "open sores in their mouths," or as the Georgia Department of Public Health (G-DPH) stated, "cervical and/or submandibular lymphadenitis and/or osteomyelitis of the mandible." The reports from the clinic indicated that "it was unlikely the infection came from contaminated equipment." The dental office then voluntarily closed, installed a water-filtration system on each dental unit, and has since reopened based on resolution of the detrimental issues. Since that report, the Georgia Public Health department has sent a letter to all providers stating that "the source of infection has been determined to be contaminated water in dental units" and raising the awareness of all professionals in the state.24,25
It boils down to having a DUWL protocol for the office to ensure the water going into a patient's mouth is clean. Having proper protocols promotes compliance. The incident in Georgia is not limited to that one office; rather, it could be a potential problem in any dental office that is not doing anything to treat the water flowing through its dental unit water lines.
Water line compliance
Successful compliance is easily implemented, cost effective, and can even reduce human error. At the onset of treatment, the use of chemicals to treat all DUWL is strongly recommended to remove as much biofilm in each DUWL line as possible. Once the initial treatment is completed, it is then advised that an automated system be immediately installed for use.26
There is a variety of chemicals that have been tested to reduce CFU levels, such as hydrogen peroxide, pure iodine, silver ions, chlorine dioxide, ozone, sodium hypochlorite, peracetic acid, and chlorhexidine gluconate. Many of these chemicals are not friendly to the environment and equipment, or can be hazardous if not used properly. It is recommended to refer to the manufacturer's instructions. The ADA states that DUWL should be cleaned and checked. If the water supply is from the city (municipal water), then a "point-of-use" cartridge should be installed or retrofitted into the unit/junction box. Bottle systems should use products that are easy, cost effective, and recognized by the EPA and FDA to confirm compliance.27,28
National Aeronautics and Space Administration (NASA) technology has been used to help prevent water contamination during dental procedures. To treat water in space, NASA incorporates an iodine resin-based technology which was developed by Umpqua Research. That technology is the same iodinated resin-bead technology found in DentaPure cartridges from Crosstex and can safely and easily be utilized in all offices; Crosstex DentaPure is rocket science for DUWL safety.29,30
DentaPure meets and exceeds ADA and CDC dental unit water quality guidelines and is EPA registered as an antimicrobial product.
DentaPure has a commitment to offering a safe, effective, and easy solution to dental unit water quality for over 17 years. DentaPure has eliminated the need to mix tablets or chemicals, requires no daily, weekly, or monthly protocols, and has no risk of leaving caustic cleaning chemicals in water lines. DentaPure is proven to be effective, and many dental and hygiene universities rely on the easy-to-use cartridges for compliance in their clinics. Feedback from college deans and directors is that DentaPure purifies water for a full year without the need to change the cartridge and extends service intervals, thereby leading to greater savings for the schools.31,32
Including DUWL safety in the office infection control protocols reduces the risk for cross contamination during patient care. In addition, by including a DUWL protocol, patients will not be concerned whether the water flowing from a handpiece, three-way syringe, or an ultrasonic cleaner is clean.
DentaPure is manufactured by Crosstex International in Hauppauge, N.Y. For more information and to see an installation video and watch the product in action, visit dentapure.com.33RDH
Ellen R. Guritzky, RDH, BS, is a respected leader, author, and speaker in the dental community. Guritzky has addressed audiences both nationally and internationally on a variety of topics such as dental prevention, health care, and business issues. In her role as the Northeast regional sales manager for Crosstex International, Guritzky proactively works with professionals and shares compliance guidelines and state standards/regulations. Prior to working with Crosstex, Ellen worked at GlaxoSmithKline and Johnson & Johnson. Ellen continues to practice in New Jersey.
References
1. US Environmental Protection Agency: www3.epa.gov/region9/water/drinking/files/dwsha_0607.pdf
2. Centers for Disease Control and Prevention: http://www.cdc.gov/healthywater/drinking/
3. CDC: http://www.cdc.gov/healthywater/drinking/public/regulations.html
4. American Public Health Association: standardmethods.org/ViewArticle.cfm?articleID=100
5. Adams OH. The Safe Drinking Water Act and the Water Utility Owner. Journal - American Water Works Association. 1977; 69 (5):229-233.
6. Centers for Disease Control and Prevention. Guidelines for Infection Control in Dental Health-Care Settings - 2003. MMWR 2003; 52(RR-17):1-68.
7. Molinari JA. Infection control: its evolution to the current standard precautions. J Am Dent Assoc 2003;134:569-574.
8. Kelsch N. Donning and removing PPE: Directions are included (CDC and OSHA). RDH Mag 2015; http://www.rdhmag.com/articles/print/volume-35/issue-5/columns/donning-and-removing-ppe.html
9. Depaola LG, Mangan D, Mills SE, et al. A review of the science regarding dental unit waterlines. J Am Dent Assoc. 1995;133:1199-1206.
10. Williams HN, Baer ML, Kelly JI. Contribution of biofilm bacteria to the contamination of the dental unit water supply. J Am Dent Assoc. 1995;126:1255-1260.
11. Coleman DC, O'Donnell MJ, Shore AC, Swan J, Russell RJ. The Role of Manufacturers in Reducing Biofilms in Dental Chair Waterlines. Journal of Dentistry 35 (2007) 701-711.
12. Molinari JA, Harte JA. Practical Infection Control in Dentistry 3rd edition. Wolters Kluwer, Lippincott, Williams & Wilkins. 2010. Pp 64-65.
13. h2odistributors.com/pages/contaminants/contaminant-heterotroph.asp
14. Wikipedia: Colony-Forming Unit: wikipedia.org/wiki/Colony-forming_unit
15. Souza-Gugelmin MCM, et al. Microbial contamination in dental unit waterlines. Braz. Dent. Journal. Scielo.Br. 14, (2003). 29 Aug. 2008 http://www.scielo.br/scielo.php?pid=S0103-64402003000100010&script=sci_arttext&tlng=en
16. CDC: http://www.cdc.gov/healthywater/drinking/public/regulations.html
17. ADA: ada.org/en/member-center/oral-health-topics/dental-unit-waterlines
18. American Dental Association: ada.org/en/member-center/oral-health-topics/dental-unit-waterlines
19. dentapure.com/wp-content/themes/dentapure/library/pdf/usaf-study.pdf
20. Cantor A. Microorganisms in Plumbing Systems. Plumbing Perspective 2016 plumbingperspective.com/microorganisms-in-plumbing-systems/
21. Microbewiki: microbewiki.kenyon.edu/index.php/Dental_Water_Line
22. thelancet.com/journals/lancet/article/PIIS0140-6736(12)60074-9/fulltext
23. medscape.com/medline/abstract/7747943
24. cbs46.com/story/30174332/infections-traced-back-to-kids-dental-clinic-parents-unaware
25. Georgia Department of Public Health - Dr. Brenda Fitzgerald, Commissioner - letter; dph.ga.gov
26. Pine P. Hidden Water Danger: What you need to know about your health and water lines. http://www.rdhmag.com/articles/print/volume-33/issue-1/featues/dental-unit-waterlines-pose-dangers.html
27. Depaola LG, Mangan D, Mills SE, et al. A review of the science regarding dental unit waterlines. J Am Dent Assoc. 2002;133:1199-1206.
28. Walker JT, Marsh PD. A review of biofilms and their role in microbial contamination of dental unit water systems (DUWS). International Biodeterioration & Biodegradation 54 (2004): 87 - 98. 28 August 2008.
29. nasa.gov/audience/foreducators/k-4/features/F_Bite_Out_of_Discomfort.html
30. spacefoundation.org/media/space-watch/space-improves-health-earth
31. nasa.gov/topics/nasalife/pure_water.html
32. dentapure.com/resources/research-science/
33. Crosstex DentaPure: dentapure.com